Review - (2023) Volume 14, Issue 3
A Review on Molecular Epidemiology of Cytomegalovirus in Northern Nigeria
Timothy Waje*,
Ediga B Agbo,
Ahmed Farouk Umar and
Iliyasu MY
Microbiology in Biological Sciences, Abubakar Tafawa Balewa University, P.M.B. 0248, Bauchi, Nigeria
*Correspondence:
Timothy Waje, Microbiology in Biological Sciences, Abubakar Tafawa Balewa University, P.M.B. 0248, Bauchi,
Nigeria,
Email:
Received: 04-Mar-2023, Manuscript No. ipacm-23-13558;
Editor assigned: 06-Mar-2023, Pre QC No. ipacm-23-13558 (PQ);
Reviewed: 15-Mar-2023, QC No. ipacm-23-13558;
Revised: 24-Mar-2023, Manuscript No. ipacm-23-13558 (R);
Published:
29-Mar-2023, DOI: 10.36648/1989-8436X.23.14.03.234
Abstract
Background: Cytomegalovirus, also known as “Human Cytomegalovirus” infects
people of all ages. The virus derives its name from its ability to cause cell
enlargement, Cyto means cell, while megalo means enlargement. It shares a
common characteristic of lifelong latency with other members of Herpesviridae
such as: Epstein Bar Virus, Varicella Zoster Virus, Herpes simplex 1 and 2, and
Kaposi Sarcoma Herpes Virus. These are associated with several illnesses such as
oral and genital blisters, congenital disorders, encephalitis, and Kaposi Sarcoma,
among others. The virus poses a significant threat to public health worldwide,
especially due to its latency and absence of effective medical treatment despite
self-limitation.
Methodology: This review covers a systematic review, meta-analysis, and scoping
review on cytomegalovirus with a view to assess the viral burden among the
studied populations and identify reported states within Northern Nigeria endemic
with the virus. The review also identified active infections and effect of the virus
on the reported people.
Results: Active pollutions with Cytomegalovirus have been reported among
pregnant women in Kebbi State (IgM 1.10%), IgM of 57.90% among blood donors,
and 7.10% in HIV patients in Sokoto State, 14.40% IgM among HIV patients with
retinitis in Kano, and 11.40%, among HIV patients, 10.50% in Pregnant women
and 23.90% among women of reproductive age respectively, in Kaduna State all in
Northwest Nigeria. Similarly, in the North, Central active infections were reported
in Benue (3.50%) among pregnant women, 19.80% among HIV patients in Bida,
and 2.60% among blood donors in Niger State, 24.90% among pregnant women in
Kwara State, 21.70% among pregnant women in Kogi State, 10.60% in HIV Children
and adolescent in the FCT Abuja, 9.50% among recurrent miscarriage women as
well as 4.80% in normal women in Plateau State respectively. Cytomegalovirus
decreases CD4 counts among HIV Patients, causes miscarriages among pregnant
women, retinitis in HIV patients, and renders blood ineligible for Donation.
Therefore, regular medical checkup and treatment of infected cases to prevent
complications are recommended.
Keywords
Prevalence; Nigeria; Molecular; Epidemiology
Introduction
Cytomegalovirus, commonly known as CMV, is an enveloped virus
with a double-stranded complex DNA molecule that encodes
over 200 viral proteins and belongs to a family of viruses called
Herpesviridae and Betaherpesvirinae subfamily [1, 2]. Moreover
called “Human Cytomegalovirus (HCMV)”, it infects people of
all ages and has a similar characteristic of lifelong latency to
other members of the family such as: Epstein Bar Virus, Varicella
Zoster Virus, Herpes simplex 1 and 2 and Kaposi Sarcoma Herpes Virus (KSHV), among others [3]. Members of the family cause a
variety of diseases including oral and genital blisters, congenital
disorders, encephalitis, and Kaposi Sarcoma [4]. Cytomegalovirus
shares the subfamily “Betaherpesvirinae” with Muromegalavirus
(infects Mice and rats), Proboscis virus (causes acute hemorrhagic
disease in elephants), Quwivirus (guinea pigs and bats as hosts),
and Roseolovirus (Mammals as host, infects T lymphocytes in
humans) [5, 6]. The Human Cytomegalovirus is spherical in shape
with a double-stranded linear DNA in an icosahedral capsid
surrounded by a lipid bilayer enveloped which composed of
various glycoproteins such as glycoprotein B (gB), gN, gH, gM, gO,
gL, and gN useful in cell attachment and penetration [7, 8] (Figure
1).
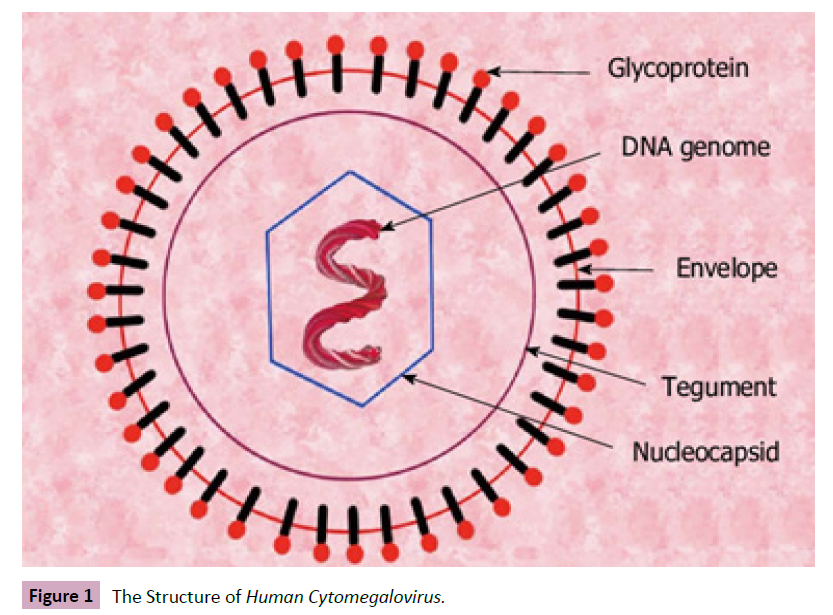
Figure 1: The Structure of Human Cytomegalovirus.
Like other viruses, the life cycle of cytomegalovirus begins with
the virion attachment to the host cells via interaction of viral
envelope glycoproteins (gH, gN, gO, etc.) with host cell surface
receptors delivering the capsid and tegument proteins into the
cell. The capsid migrates to the nucleus where the viral genome
is freed. The tegument proteins regulate host response as well as
genome replication in the order of early and immediate (IE 1) gene
expression, delayed early (IE 2) gene expression, and late gene
expression, respectively. The late gene expression initiates capsid
assembly in the nucleus, which associates with the tegument
proteins and migrates to the viral assembly complex where they
acquire further tegument and envelope (virions encapsulation of
replicated DNA as capsids) .These migrate to the cytoplasm where
secondary development occurs at the Endoplasmic Reticulum
and Golgi Apparatus before being release together with dense
non-infectious agents by exocytosis [9, 10] (Figure 2).
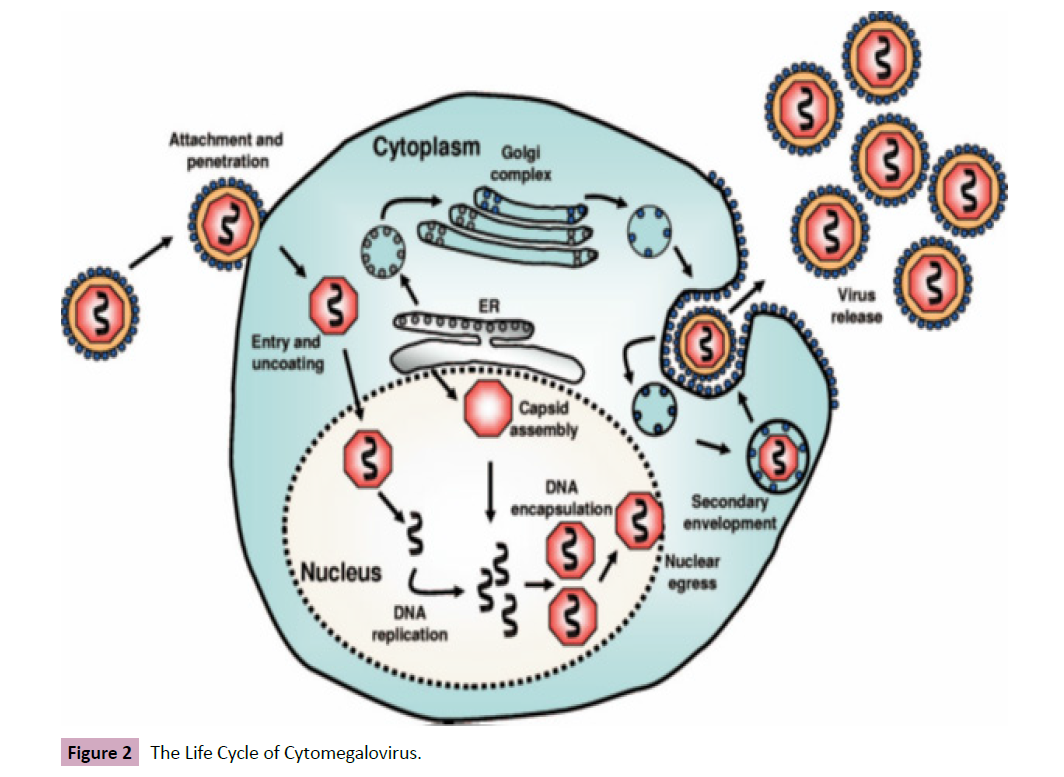
Figure 2: The Life Cycle of Cytomegalovirus.
Genetic diversity has been reported in Cytomegalovirus through
genotyping of the viral glycoproteins gB1, gN4AN1, gB3, gH2, gB2,
gB4,) from PCR positive clinical samples (urine, saliva, and plasma) producing gB2, gN3BN1, gB1, 2, 4, gH1, gB1, gB2 subtypes among
others. The diversity in viral glycoproteins plays a great role in
viral tropism and spread. Mutated genes such as UL9, UL1, RL6,
RL5A, and US9 due to deletions and insertions in Cytomegalovirus
genome have been reported with shared ancestral features.
Virulence genes such as UL55 (codes for gB for attachment ) and
US9 (codes gpUS9 cell-to--to-cell spread in epithelia cells) play a
key role in virus tropism while UL83 ( codes for matrix protein,
inhibits antigen processing of 72-kDa IE protein), US3 ( gp32/33,
retains Major Histocompatibility Complex(MHC) class 1 molecules
loaded with peptides in Endoplasmic Reticulum ER) and US2/
US11 (gp24/gp33, directs detained MHC class 1 molecules from
ER to proteasome for degradation) are genes that enable the
virus to evade the host immunity.
The Human Cytomegalovirus is described as the most successful
human viral pathogen due to its transmission routes which are
utero-perinatal and postnatal with the ability to remain latent
in the host for a very long time without evoking clinical signs
and symptoms. It can be activated by critical illness, drugs,
inflammatory and stress mediators, immunosuppression, organ
transfer, among other factors. The virus has been reported
among women within child bearing age posing a significant risk
for perinatal or neonatal transmission. The common mode of
transmission of the virus is through direct contact of soft tissues
or open sores with body fluids of infected persons such as blood,
tears, urine, breast milk, semen, saliva, and infant contact with the
mother’s genital secretions during birth. The virus spreads within
an individual causing multi-systemic effect involving many tissues
and organs such as lungs, liver, eye (retina), muscles, brain, and
gut among others [10]. Although latency results in asymptomatic
infections, viral activation leads to symptoms such as fever,
encephalitis (brain inflammation) causing seizures and coma, pneumonia with hypoxemia, liver inflammation (hepatitis), large
ulcers, shortness of breath, and vision problems. Cytomegalovirus
can cause sore throat, fatigue, hepatitis, mononucleosis, and
swollen glands [3]. Complications can result from gastrointestinal
problems (diarrhea, colon inflammation, abdominal pain, and
blood in the stool), pneumonitis, and inflammation of the brain
(encephalitis), and liver problems. Immunocompromised persons
such as HIV patients and organ transplant recipients are at
serious risk for Cytomegalovirus contraction. Infected pregnant
women pass the virus to either the unborn child in the womb
via blood circulation or through contact of the baby with genital
secretions during delivery. Cytomegalovirus can be diagnosed in
the laboratory by the following ways:
1. Serologic investigations for IgM in active infection and IgG
due to a previous exposure/recovery.
2. Detection of viral antigen or DNA using Polymerase Chain
Reaction (PCR).
3. Culture of urine samples particularly in infants.
4. Biopsy in immunocompromised patients.
Cytomegalovirus infection is self-limiting with occasional recovery
without medication, especially in adults, but medications
(antivirals) can be administered to children and adults with
weak immunity to slow viral replication. Although Ganciclovoir
may have adverse effects, it reduces viral spread in those with congenital or recurring CMV infections, so organ damage may
require hospitalization. Cytomegalovirus is a public health concern
globally. Therefore, this paper involves a systematic review, metaanalysis,
and scoping review with the aim of investigating the
molecular epidemiology of cytomegalovirus in Northern Nigeria.
Epidemiology of Human Cytomegalovirus
Epidemiology is the study of the distribution (frequency and
pattern) and determinants in relation to causes and risk factors
for health-related issues and diseases in a specified population
with application for prevention and control. Several factors
are involved in the epidemiology of Human Cytomegalovirus.
Although the virus is highly prevalent globally, its prevalence
has a direct correlation with socio-economic status and advance
age. Organ recipients, persons with weakened immunity, and
those on immunosuppressive drugs are at a high risk of CMV
contraction. A CMV prevalence of 94.50% was reported among
persons with platelet disorders, 83.30% in those with anemia and
leukemia, and 91.00% in a cross-sectional study among patients
with hematological diseases in the Western Brazilian Amazon.
A high prevalence of the virus has been reported in developing
countries where the socio-economic status are low compared
to Europe, some parts of Australia, and North America with low
prevalence. Human Cytomegalovirus Immunoglobulin G (IgG)
prevalence of 39.30% and 48.0% was reported among adult men
in France and United States, respectively, but among women of reproductive age this was 45.60%-95.70% (Europe), 60.20%
(Japan), 58.30%-94.50% (LATAM), and 24.60%-81.0% (North
America. Maternal and congenital CMV Seroprevalence of 98.1%
and 8.4%, respectively, was reported among mothers and infants
in Columbia. Genetic variability in Human Cytomegalovirus has
been described as complex and enables the virus access to
multiple hosts with the ability to thrive well even in unfavorable
conditions. This diversity in Human Cytomegaloviruses has been
linked to changes in the envelope glycoproteins (gM, gO. gN, gH,
etc) producing subtypes. Polymorphism (variants) of HCMV was
reported in UL54 (P342S, S384F, K434R, S673F, T754M, R778H,
C814S, M827I, G878E, S880L, E888K and S976N) and in UL97
(M615T) among DNAemia patients in Taiwan. In a Phylogenetic
analysis of UL146, UL144, and UL55 genes, distinct strains of
HCMV were reported among different families.
In Africa, Human Cytomegalovirus is a common etiologic agent
of congenital infections in children as well as pneumonia and
meningitis, particularly among immunosuppressed hospitalized
patients. The endemicity has been described high in young infants
18 months of age with a prevalence rate of 83.00% in Zambia
while in Nigeria high prevalence among prospective blood donors
was significantly associated with overcrowding. The progression
in the burden of HIV in Africa with its increasing morbidity
and mortality has been associated with the effect of Human
Cytomegalovirus either as a secondary pathogen or co-pathogen with HIV exerting a concerted effects against the immune system
of their host with a consequent inflammatory effects.
Human Cytomegalovirus in Nigeria
Nigeria consists of 36 states and the Federal Capital Territory (FCT)
distributed in six geopolitical zones such as North West, North
East, North Central, South-South, South West and South East
respectively. Northern Nigeria is made up of three geopolitical
zones which include: North West, North East and North Central
also known as Middle Belt (Figure 3).
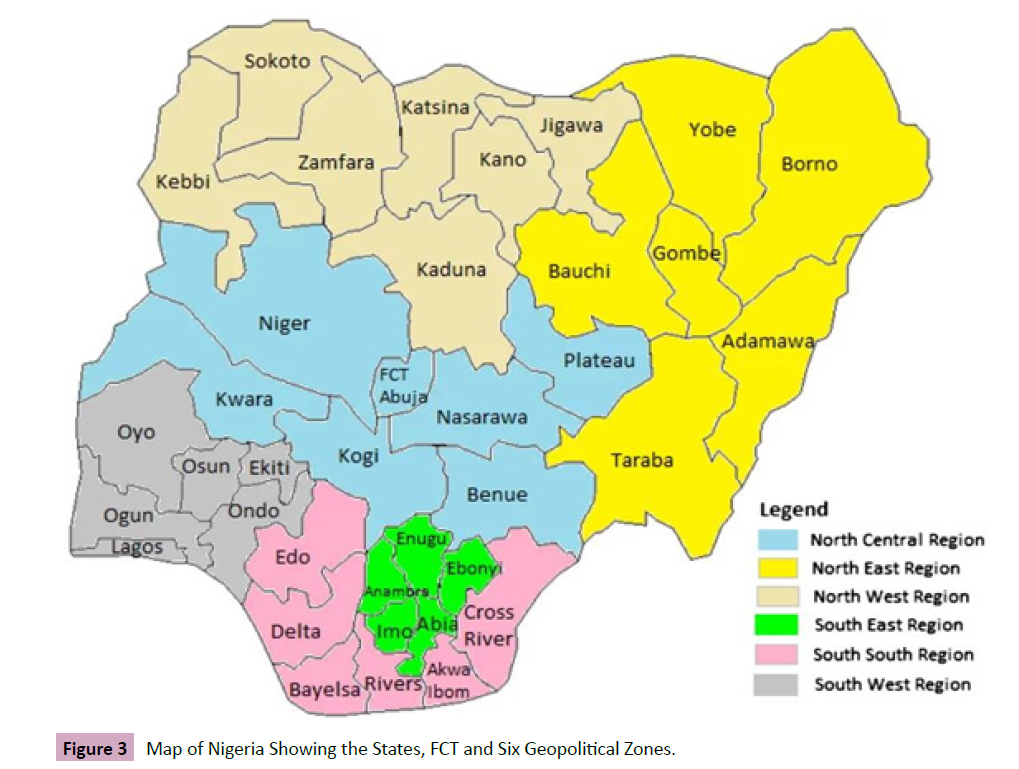
Figure 3: Map of Nigeria Showing the States, FCT and Six Geopolitical Zones.
There have been several reports on the prevalence of Human
Cytomegalovirus in Nigeria. Anti-HCMV IgG prevalence rate of
86.00% and 13.20% for IgM among HIV patients with 72.00%
IgG with 2.80%IgG in HIV negative persons has been reported in
Nigeria. A prevalence rate of 3.80% was reported among infants
in Lagos through Polymerase Chain Reaction with two positive
symptomatic subjects with liver and spleen enlargement and
pneumonia related illness. Furthermore, a Seroprevalence of
97.2% of IgG against Cytomegalovirus was reported among
normal pregnant women in Lagos, Nigeria. In a related study,
a prevalence of 60.00% IgG to the virus was reported among
pregnant women in Oshogbo Ogun State. In Abakaliki Ebonyi
State, a HCMV infection prevalence of 11.30% was reported
among HIV positive women and 16.70% of HIV positive pregnant
women.
In North West Nigeria, 1.1% anti-CMV IgM was reported among
pregnant women in ante-natal clinics in Birnin-Kebbi, Kebbi State
while 93.30% with IgG antibodies in a related report. An anti-CMV
IgG of 97.80% was reported among pregnant women attending
Usman Danfodiyo University Teaching Hospital (UDUTH) in
Sokoto State. Similarly, a prevalence of 4.82% IgG and 57.90 IgM
CMV antibodies was reported among blood donors and 16.60%
IgG with 7.10% IgM CMV antibodies among HIV positive patients
in Sokoto. An anti-CMV IgG of 91.10% was reported among
pregnant women attending Murtala Mohammed Specialist
Hospital (MMSH), Kano, Nigeria. Similarly, 100.00% prevalence of
anti-HCMV IgG and PCR Viraemia of 14.40% in HIV infected adults
with retinitis attending Aminu Kano Teaching Hospital (AKTH)
was reported in Kano, Nigeria. In Kaduna State, anti-HCMV IgG
prevalence of 99.40% and IgM of 11.40% was reported among
HIV patients (in Zaria, Kwoi, and Kaduna Metropolis) while a CMV IgG antibody prevalence of 94.80% was reported among pregnant
women with 100% IgG antibody prevalence in non-pregnant
women attending selected hospitals in Kaduna State. Similarly
anti-HCMV IgM of 10.50% was reported among pregnant women
attending ante-natal clinic at the General Hospital Kafanchan
(GH Kaf), Kaduna State. Furthermore. In Zaria Kaduna State, anti-
HCMV IgG of 94.70% and IgM of 23.90% was reported among
women of reproductive age in selected hospitals (Table 1).
| S/N |
State |
Location |
Test Type |
IgG Prevalence Rate (%) |
Active Infection/IgM Prevalence Rate (%) |
Population |
| i. |
Kebbi |
Birnin Kebbi |
ELISA |
NRS |
1.1 |
Pregnant Women- |
|
|
Birnin Kebbi |
ELISA |
93.3 |
NRS |
Pregnant Women |
| ii. |
Sokoto |
UDUTH, Sokoto |
ELISA |
97.8 |
NRS |
Pregnant Women |
|
|
Sokoto |
ELISA |
4.82 |
57.9 |
Blood Donors |
|
|
Sokoto |
ELISA |
16.6 |
7.1 |
HIV Patients |
| iii. |
Zamfara |
NRS |
NRS |
NRS |
NRS |
NRS |
|
Katsina ,Jigawa |
|
|
|
|
|
| iv. |
Kano |
MMSH Kano |
ELISA |
91.1 |
NRS |
Pregnant Women |
|
|
AKTH Kano |
ELISA/PCR |
100 |
14.4 |
HIV Patients with Retinitis |
|
|
|
ELISA |
98 |
NRS |
Pregnant Women |
| v. |
Kaduna |
Zaria, Kwoi, Kaduna |
ELISA |
99.4 |
11.4 |
HIV Patients |
|
|
Kaduna |
ELISA |
94.8 |
NRS |
Pregnant Women |
|
|
Kaduna |
ELISA |
100 |
NRS |
Non -Pregnant Women |
| vi. |
|
Kafanchan |
ELIZA |
NRS |
10.5 |
Pregnant Women |
| vii. |
|
Zaria |
ELISA |
94.7 |
23.9 |
Women of Reproductive Age |
Table 1. Cytomegalovirus in Northwest Nigeria.
Keys=ELISA-Enzyme-Linked Immuno Sorbent Assay, NRS=No Report Seen, PCR=Polymerase Chain Reaction
In the North Central/Middle Belt, a prevalence of 93.30%
anti-CMV IgG seropositive and anti-CMV IgM of 3.50% with a
significant association with gravidity. In pregnant women was
reported in Makurdi, Benue State. Similarly, anti-CMV IgM
prevalence of 19.80% among HIV patients in Bida and 96.20% IgG
with 2.60% IgM among blood donors in Minna, Nigeria State. In
Kwara State, a CMV IgG prevalence of 98.20% and IgM of 24.90%
was reported among pregnant women in Ilorin. A prevalence of anti-HCMV IgM of 21.70% was reported among pregnant women
in Lokoja, Kogi State. A prevalence of anti- HCMV IgG of 74.00%
was reported among eligible blood donors in Keffi, Nasarawa
State, while 10.60% anti-HCMV IgM among HIV infected Children
and Adolescents in the Federal Capital Territory (FCT) Abuja,
respectively. A Seroprevalence of CMV IgG of 85.70% and 9.50%
IgM antibodies among women with recurrent miscarriages and
76.20% IgG and 4.80% IgM antibodies among normal women was
reported in Jos Plateau State. In the North East, while no literature
was located through various search engines for the spread
of Cytomegalovirus in Taraba, Gombe, Bauchi, and Adamawa
States as at the time of this report. A 100.00% anti-CMV IgG was
reported among HIV positive and 98.60% in HIV negative patients
attending University of Maiduguri Teaching Hospital (UMTH) in
Borno State, An anti-CMV IgG prevalence of 90.00% was reported
among pregnant women attending ante-natal in Nguru in Yobe
State out of which 92.80% had still birth and 86.6% miscarriages
(Table 2).
| S/N |
State |
Location |
Test Type |
IgG Prevalence Rate (%) |
Active Infection/IgM Prevalence Rate (%) |
Population |
| i. |
Benue |
Makurdi |
ELISA |
93.3 |
3.5 |
Pregnant Women |
| ii. |
Niger |
Bida |
ELISA |
NRS |
19.8 |
HIV Patients |
|
|
Minna |
ELISA |
96.2 |
2.6 |
Blood Donors |
| iii. |
Kwara |
Ilorin |
NRS |
98.2 |
24.9 |
Pregnant Women |
| iv. |
Kogi |
Lokoja |
ELISA |
NRS |
21.7 |
Pregnant Women |
| v. |
Nasarawa |
Keffi |
ELISA |
74 |
NRS |
Blood Donors |
| vi. |
FCT |
Abuja |
ELISA |
NRS |
10.6 |
HIV Children and Adolescents |
| vii. |
Plateau |
Jos |
ELISA |
85.7 |
9.5 |
Recurrent Miscarriages Women |
|
|
Jos |
ELISA |
76.2 |
4.8 |
Normal Women |
| viii. |
Gombe, Bauchi, Taraba and Adamawa |
NRS |
NRS |
NRS |
NRS |
NRS |
| ix. |
Borno |
UMTH |
ELISA |
100 |
NRS |
HIV Positive |
|
|
UMTH |
ELISA |
98.6 |
NRS |
HIV Negative |
| x. |
Yobe |
Nguru |
ELISA |
90 |
NRS |
Pregnant Women |
|
|
Nguru |
ELISA |
92.8 |
NRS |
Women with Still Birth |
|
|
Nguru |
ELISA |
86.6 |
NRS |
Miscarriage Women |
Table 2. Cytomegalovirus in North Central and North East, Nigeria.
There was little literature on the molecular study of Human
Cytomegalovirus in Northern Nigeria at the time of compiling
this report. Nevertheless, Cytomegalovirus glycoprotein B gene
(gB) detected through PCR was reported among anti-CMV IgM
positive prevalence of 11.40 in HIV patients in Kaduna. Similarly,
Glycoprotein gB as well as variable genes gB1 (83.70%) and
gB2 (16.30%), detected through PCR was reported among anti-
CMV IgM prevalence of 19.80 in HIV patients in Bida and 2.60%
among blood donors in Minna all in Niger State. Cytomegalovirus
decreases CD4 counts and contributes to retinitis among HIV
patients, miscarriage in pregnant women and renders blood
donors ineligible (Table 3).
| State |
Location |
Population |
IgM Prevalence |
Glycoprotein Gene |
Variable Gene |
Effect of Cytomelavirus |
| Kaduna |
Kaduna |
HIV Patients |
11.4 |
gB |
NRS |
NRS |
| Niger |
Bida |
HIV Patients |
19.8 |
gB |
gB1, gB2 |
Deceases CD4 Counts |
|
Minna |
Blood donors |
2.6 |
NRS |
NRS |
Renders blood ineligible for donation |
| Kano |
Kano |
HIV Patients |
14.4 |
NRS |
NRS |
Decreases CD4, Causes Retinitis |
| Plateau |
Jos |
Pregnant |
9.5 |
NRS |
NRS |
Recurrent Miscarriages |
| Yobe |
Nguru |
Pregnant |
NRS |
NRS |
NRS |
Miscarriages in Women |
|
|
Women |
|
|
|
|
|
Nguru |
Pregnant Women |
NRS |
NRS |
NRS |
Still Birth |
Table 3. Genetic Variability, Effects of Human Cytomegalovirus in Northern Nigeria.
Conclusions and Recommendations
This review confirms previous reports that cytomegalovirus
poses a significant threat to public health globally. The virus
infects Immuno-compromised persons with complications
development. The presence of immunoglobulin g (IgG) signifies passed infection while IgM is an active infection. Active infections
with Cytomegalovirus have been reported among pregnant
women in Kebbi State (IgM 1.10%), IgM of 57.90% among blood
donors, and 7.10% in HIV patients in Sokoto State, 14.40% IgM
among HIV patients with retinitis in Kano, and 11.40%, among
HIV patients, 10.50% in Pregnant women and 23.90% among
women of reproductive age respectively, in Kaduna State all
in Northwest Nigeria. Similarly, in the North Central, active
infections were reported in Benue (3.50%) among pregnant
women, 19.80 among HIV patients in Bida, and 2.60% among
blood donors in Niger State, 24.90% among pregnant women
in Kwara State, 21.70% among pregnant women in Kogi State,
10.60% in HIV Children and adolescent in the FCT Abuja, 9.50%
among recurrent miscarriage women as well as 4.80% in normal
women in Plateau State respectively .There is no report of
active infections in the whole of the North Eastern States Such
as Gombe, Bauchi, Adamawa and Taraba States respectively but
passed infections with IgG reports in Borno State (100% and 98%)
and Yobe State (90%, 92.80% and 86.60%) respectively. Therefore,
cytomegalovirus has been reported in Northern Nigeria among
HIV patients, pregnant women, and blood donors at different
locations within the region. The virus has been associated with
decreased CD4 count among HIV patients, recurrent miscarriages,
and still births among pregnant women, renders blood ineligible
for donation, and is implicated in retinitis among HIV patients.
Therefore, regular medical checkups, avoidance of exposure of
soft tissues and open sores to infected body fluids, and medical
attention to infected cases to avoid complications and viral
spread are recommended for proper control of the virus. More
study is recommended in Gombe, Adamawa, Bauchi and Taraba
States respectively. Adequate viral control is necessary for the
improvement of public health.
Conflict of Interest
All authors declare no conflict of interest, financial or nonfinancial.
References
- Bodaghi B, Michelson S (2009) Cytomegalovirus Virological Facts for Clinicians. Ocur Imm and Inflam 3: 133-137.
Indexed at, Google Scholar, Crossref
- Luangsirithanya Panupong, Treeware Sukrit, Pongpaibul Ananya, Pausawasdi Nonthalee, Limsrivilai Julajak, et al. (2021) Cytomegalovirus enterocolitis with Subsequent Diagnosis of Coexisting new onset bowel disease; two case reports and review of the literature.Clin Case Rep 100: 1-8.
Indexed at, Google Scholar, Crossref
- Centers for Diseases Control and Prevention (CDC) (2020) Cytomegalo Virus and Congenital CMV infection.
- John E, Bennett (2020) Introduction to Herpesviridae in Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. Enceclopedia of Virology 10: 303-304.
Google Scholar, Crossref
- MicrobeWiki (2010) Herpesviridae. Baltimore Classification.
Google Scholar
- Michael A Purdy, Jan Felix Drexler, Xiang-Jin Meng, Heléne Norder, Hiroaki Okamoto, et al. (2022) International Committee on Taxonomy of Viruses Herpesviridae. J Gen Viro 103.
Indexed at, Google Scholar, Crossref
- Microbe Wiki (2022) Human Cytomegalovirus. Transfus Med Hemother 37: 365-375.
Google Scholar, Crossref
- Jasbir S Makker, Bharat Bhajantri, Sailaja Sakam, Sridhar Chilimuri (2016) Cytomegalovirus related fatal duodenal diverticular bleeding: a case report and literature review. World J Gastroenterol 22: 7166-7174.
Indexed at, Google Scholar, Crossref
- Tania Crough, Tajiv Khanna (2009) Immunobiology of Human Cytomegalovirus: from Bench to Bedside. Clinical Microbiology Reviews. Research Gate 22: 76-98.
Indexed at, Google Scholar, Crossref
- Pierre M, Jean Beltran, Ileana M Cristea (2014) the Life Cycle and Pathogenesis of Human Cytomegalovirus Infection: Lessons from Proteomics.Expert Rev Proteom 11: 687-711.
Indexed at, Google Scholar, Crossref
Citation: Waje T, Agbo EB, Umar AF, Iliyasu MY, (2023) A Review on Molecular Epidemiology of Cytomegalovirus in Northern Nigeria. Arch Clinic Microbio, Vol. 14 No. 3: 234.








