Keywords
|
| Butea monosperma; Antiepileptic activity; Maximal electroshock; Pentylenetetrazole; Pentobarbital |
Introduction
|
| Traditional, alternative or herbal medicine refers to medical/ medication practices other than orthodox medicine/medical practice [1]. The WHO has reported that about 80% of people in the third world countries depend wholly or partly on herbal medicines for their general medications [2]. Many researchers have tried to establish a scientific link/basis to explain the traditional claims of efficacy of most herbs by using different in vitro and in vivo experimental models. Neurological disorders such as Epilepsy, Parkinson disease, Alzheimer disease, anxiety, depression and other psychiatric disorders have been studied extensively [3-5]. The present study aims to investigate the anticonvulsant activity of methanol stem extract of Butea monosperma (Lam.) Kuntze (BMME) and its isoalted bioactive compound by using the Maximal electroshock (MES) convulsion and the Pentylenetetrazole (PTZ) induced convulsion models [6]. |
| The plant Butea monosperma (Lam.) Kuntze belongs to the family Fabaceae. The leaves are believed to have astringent, depurate, diuretic and aphrodisiac properties. It promotes diuresis and menstrual flow. The seed is having anthelmintic property. The bark is also used in snakebite as antivenom. Its seeds are when pounded with lemon juice and applied to the skin, it acts as a rubefacient [7]. The main constituents of this plant are butrin, butein, butin, Terpines such as lupeol and lupenone. Butea monosperma (Lam.) Kuntze also contains flavonoids and steroids. Flowers containcoreopsin, isocoreopsin, sulphurein, monospermoside and isomonospermoside were also identified [8]. |
| Epilepsy is a neurological disorder characterized by recurrent convulsions of cerebral origin, with episodes of sensory, motor or autonomic phenomenon with or without loss of consciousness. About 10% of the patients are refractory and continue to have convulsions at interval of one month or less, which severely disrupts their life and work [9]. Epilepsy is the third most common neurological disorder in the US after Alzheimer’s disease and Stroke. Its prevalence is greater than Cerebral palsy, Multiple sclerosis and Parkinson’s disease combined [10]. Antiepileptic drugs (AEDs) currently in use notably Phenytoin, Carbamazepine, Phenobarbital and Valproate, referred to as older AEDs were (drugs introduced before 1990). These drugs induce hepatic microsomal enzymes, cytochrome P450 (CYPs) thereby complicating the use of multiple anti convulsion drugs as well as impacting metabolism of oral contraceptives, warfarin and many other drugs. The drugs also enhance metabolism of endogenous compounds including gonadal steroids and vitamin D [11]. These short comings has led to a renewed reawakening of interest in the search for medicinal plants and their active component that will be effective for the management of diseases including epilepsy. |
Materials and Methods
|
|
Plant collection
|
| Stem of Butea monosperma (Lam.) Kuntze was collected from the forest area of Midnapur district, West Bengal, India. The parts were shade dried and herbarium was prepared for the authentication. The powdered stems of Butea monosperma (Lam.) Kuntze was authenticated from National Bureau of Plant and Genetic Resources (NBPGR) (Ref. No. NHCP/NBPGR/2010/17), New Delhi. Further, phytochemical investigations was undertaken to know the presence of phytoconstituent in the raw material of Butea monosperma (Lam.) Kuntze. |
|
Extraction
|
| Dried stem powder of Butea monosperma (Lam.) Kuntze (70 g) was exhaustively extracted with 500 mL petroleum ether and then with methanol in Soxhlet apparatus for 48 h. A dark brown residue (9.8 g) was obtained after evaporation of the solvent. The stem extract of Butea monosperma (Lam.) Kuntze was used for the further studies. |
|
Animals
|
| Swiss Albino mice of either sex weighing 25-35 g, 3-4 months of age, were used for the experiment. Animals were housed in group of 6 per cage and maintained in the animal house. They were obtained from Central animal house of Noida Institute of Engineering and Technology (NIET), Greater Noida, Uttar Pradesh. They were given ad libitum water and standard pellet diet under strict hygienic conditions. Animals were acclimatized to laboratory conditions before testing e.g., room temperature of 25 ± 1°C, relative humidity 45-55% and a 12:12 h light dark cycle. |
| The experimental protocol was approved from the Institutional Animal Ethics Committee (IAEC) and protocol approval No. is NIET/ IAEC/2009/06. The Committee for Purpose of Control and Supervision of Experiments on Animals (CPCSEA) Reg. No. is 1121/ac/07/ CPCSEA. All the experimental procedures were carried out according to the proper guidelines of CPCSEA. |
|
Drugs/chemicals and treatment
|
| Pentobarbitone (Sigma Aldrich, Bangalore), Pure Diazepam was obtained as gift sample from Cipla, Mumbai, Pentylenetetrazole (PTZ) and Carboxy Methyl Celllulose (CMC) were obtained from Sigma Aldrich, Bangalore and CDH, Mumbai respectively. All other chemicals used were of analytical grade. |
|
Phytochemical screening
|
| Crude methanol extract of Butea monosperma was screened for the presence of secondary metabolites such as alkaloids, tannins, saponins, glycosides, flavonoids, steroids and triterpenoids. The screening was done using standard protocol described by Trease and Evans [12,13]. |
|
Acute toxicity studies
|
| The acute oral toxicity study was carried out according to OECD guidelines 423 [14]. First one animal is dosed with 100 mg/kg body weight. If animal dies a much lower dose is tested. If animal survives, then two more animals are dosed, after 48 hrs observation of the first animal. If survives, then the main test should be terminated. If animal dies, two more animals are dosed and observed. The study was performed with an initial dose of 100 mg/kg body weight. The dose sequence followed were 100, 200, 500, 1000 and 2000 mg/kg body weights. |
|
Pentobarbital-induced sleep in mice
|
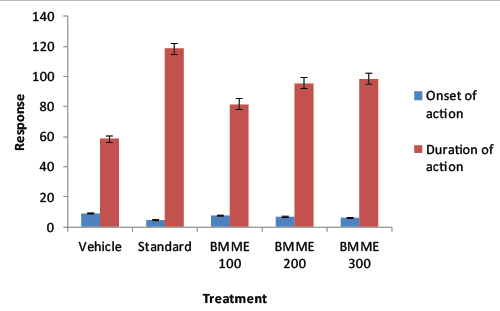
| Swiss albino mice of either sex were divided into five groups of six mice each. Group I served as vehicle control, group II served as standard group, group III, IV and V served as test groups (BMME was given in 100, 200 and 300 mg/kg p.o.). Thirty minutes after treatment all the groups were administered with pentobarbital 45 mg/kg i.p. The onset and duration of sleep was observed and recorded with the mice placed in individual cages. Loss of rightening reflex was considered as the criterion for sleep [15] while the interval between the loss and the recovery of straightening was regarded as the duration of sleep [16]. |
|
Pentylenetetrazole (PTZ) induced convulsion in mice (Chemically-induced convulsion model)
|
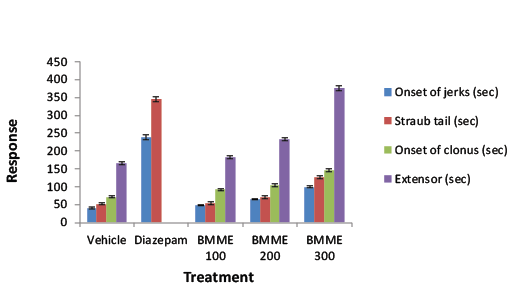
| Convulsion was induced in mice by administration of PTZ (80 mg/ kg i.p.). Jerky movement, straub tail, clonus, extensor were recorded. The animals were divided into 5 groups of 6 animals each [17]. |
|
Maximal electroshock convulsion (MES) in mice
|
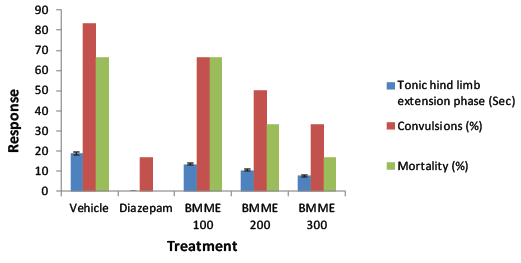
| Convulsion was induced in the animals by electroconvulsive shock (50 mA for 0.2 s) via a pinna electrode using an electroconvulsiometer. The duration of hind limb tonic extension (HLTE) was compared with animals of control group. Decrease in the duration of hind limb extension was considered a protective action. Animals shown convulsion and mortality were also recorded. Less convulsion and mortality indicated positive action. The animals were divided into 5 groups of 6 animals each. Group I served as control (vehicle) group (treated with 1% w/v carboxy methyl cellulose, 0.25 ml, p.o.). group II was the reference group and was treated with diazepam (4 mg/kg i.p. 30 min) prior to the induction of convulsion. Groups III, IV and V served as test groups and were treated with the BMME (100, 200 and 300 mg/ kg p.o. respectively) for 7 days [17]. |
|
Isolation of compound from the methanol extract of stem of Butea monosperma (Lam.) Kuntze
|
| The stem extract of Butea monosperma (Lam.) Kuntze (100 g) was dissolved in methanol and centrifuged at 2000 rpm for 30 min. The residue was placed on top of the silica gel column (60-120 mesh) and eluted by column chromatography with chloroform : methanol (95:05 v/v), it was further re-chromatographed on silica gel (100–200 mesh) column chromatography resolved into a pure compound eluted by chloroform: methanol (50:50, v/v) [18]. |
|
Statistical analysis
|
| Results were expressed as mean ± standard error of mean. Statistical analysis was performed by analysis of variance (ANOVA); when a statistically significant result was obtained with ANOVA, a Dunnets t-test was performed for multiple comparisons. Values of p<0.05 were considered significant. |
Results
|
|
Acute toxicity studies
|
| According to the OECD guidelines 423, the animals were observed individually after respective dosing, at least once during first 30 min periodically during the first 24 h, and thereafter, for 2 days. Observation includes changes in fur, skin, eye, respiratory, circulatory, central nervous System, mucous membrane, motor activity, behavioural changes, convulsions, salivation, tremors, sleep and mortality were observed. The treated animals did not show any symptoms of toxicity. Hence, BMME was considered to be safe upto 2 g/kg bw. Based upon these, three doses of BMME were selected i.e., high dose (300 mg/kg; bw), medium dose (200 mg/kg; bw) and low dose (100 mg/kg; bw) for in vivo pharmacological experiments (Tables 1-4) (Figures 1-3). |
|
Isolation and characterization
|
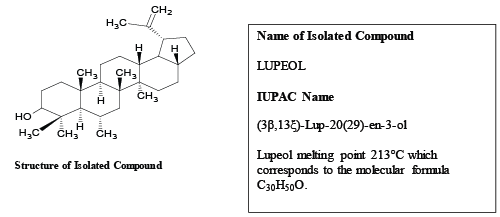
| Isolation of bioactive molecule from the methanol stem extract of stem of Butea monosperma (Lam.) Kuntze: The isolated compound was obtained from the methanol extract of stem of Butea monosperma (Lam.) Kuntzeas white crystals. The name had given to it was TBM. |
|
Spectral analysis
|
| FTIR finger print analysis of isolated compound from the methanol stem extract of Butea monosperma (Lam.) Kuntze: The FTIR spectra was interpreted as follows IR (KBr): 3384.58 cm-1 (Hydrogen bonded OH Stretch), 2942.36 cm-1 (C-H Stretch in CH2), 1645.85 cm-1 (C=C Symmetric Stretch), 1485.69 (C=C Asymmetric stretch), 1463.19 cm-1(C-H deformation in CH2 and CH3), 1388.10 cm-1(C-H Stretch), 1027.79 cm-1 (C-O Stretch of secondary alcohol), 882.87 cm-1(=C-H bending exocyclic CH2). |
|
NMR spectral study of isolated compound from the methanol stem extract of Butea monosperma (Lam.) Kuntze
|
| a) 1H NMR peak of isolated compound from the methanol stem extract of Butea monosperma (Lam.) Kuntze: The 1H NMR: 7.260 (CDCL3 peak), 4.683, 4.565(H-29, d, d, 2H), 3.202-3.194 (H, 3, d, d, 1H, 6 Hz, 5 Hz), 2.392(H-19, m, 1H), 2.381 (H-21a, m, 1H), 2.360 (H-15A, t, 1H), 2.348 (H-30, s, 3H), 1.609 (H-12A, 1A, d, 2H), 1.458 (H-13, t, 1H), 1.322 (H-2A, d, 1H), 1.232 (H-2B, q, 1H), 1.198 (H-12A, q, 1H), 1.176 (H-23, s, 3H), 1.080 (H-15A, d, 1H), 0.966 (H-23, s, 3H), 0.942 (H-27, s, 3H), 0.905 (H-18, t, 6 Hz, 1H), 0.787 (H-28, s, 3H), 0.759 (H- 24, s, 3H), 0.690 (H-25, s, 3H), 0.671 (H-5, d, 1H). |
| b) 13CNMR peak of isolated compound from the methanol stem extract of Butea monosperma (Lam.) Kuntze: In the 13C NMR spectrum of Lupeol showed δC: δ 37.354 (C-1), δ 21.108 (C-2), δ79.178 (C-3), δ38.241 (C-4), δ55.481 (C-5), δ18.171 (C-6), δ27.626 (C-7), δ38.886 (C-8), δ50.625 (C-9), δ34.463 (C-10), δ19.476 (C-11), δ21.108 (C-12), δ35.760 (C-13), δ39.031 (C-14), δ25.334 (C-15), δ29.857 (C-16), δ40.176 (C-17), δ48.493 (C-18), δ48.159 (C-19), δ151.135 (C-20), δ27.596 (C-21), δ38.886 (C-22), δ25.334 (C-23), δ15.523 (C- 24), δ16.145 (C-25), δ15.98 (C-26), δ14.719 (C-27), δ16.282 (C-28), δ109.477 (C-29) and δ18.490 (C-30). |
| Mass spectra of isolated compound from the methanol stem extract of Butea monosperma (Lam.) Kuntze: The bioactive isolated compound from BAME had given molecular ion peak [M+1] 425.379 in ESI mass spectrum suggested molecular formula C30H50O. |
| On the basis of phytochemical screening the isolated compound was found to be a Triterpene. The spectral data of FTIR, NMR and MASS were interpreted to predict the molecular structure, atomic stretching, possible molecular functional groups, etc. The compound thus interpreted was lupeol, a triterpenoid, isolated compound from stem of Butea monosperma (Lam.) Kuntze (Figures 4 and 5). |
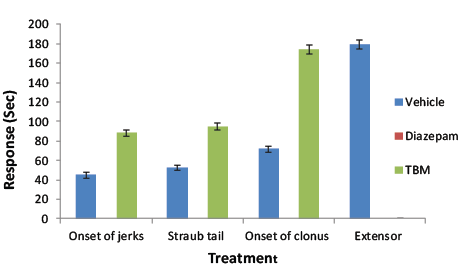
| The isolated compound, TBM from BMME was screened to establish its effect on PTZ induced convulsion. In PTZ induced convulsions onset time of jerk, straub tail, clonus and extensor were significantly increased. The results were better enough than the effects given by BMME on the respective parameters (Table 5). |
Discussion
|
| Phytochemical screening of the extract revealed the presence of secondary metabolites such as terpenoids, tannins, glycosides and flavonoids. Anticonvulsant effect of saponins and flavonoids has been reported [19,20]. The sub acute toxicity study showed no sign and symptoms of toxicity. The extract dose dependently reduced the onset of pentobarbital induced sleep, pentobarbital acts by increasing GABA mediated synaptic inhibition either by directly activating GABA receptors or, more usually, by enhancing the action of GABA on GABAA receptors [9]. The total sleep time was potentiated significantly at the dose 300 mg/kg suggesting that the extract of Butea monosperma has sleep potentiating properties [21]. |
| Butea monosperma extract produced 50-100% protection of the mice against PTZ induced convulsion at doses of 100-300 mg/kg. The protection of the extract against PTZ induced convulsion suggested that the extract interacts with GABA-ergic neurotransmission. The PTZ test is assumed to identify anticonvulsant drugs effective against myoclonic and absence convulsions. Butea monosperma significantly attenuated electrically induced convulsion in mice. Electroshock convulsions are characterized by tonic extension of the hind limb and abolition of this activity is taken as anticonvulsant action. The ability of Butea monosperma to decrease the duration of tonic clonic convulsion in the MES test shows its activity against generalized tonic clonic convulsions [22-25]. Butea monosperma has demonstrated potent activity against PTZ and MES convulsions and it would generally be right to say that it will be effective against absence convulsions and generalized tonicclonic convulsions. Since the MES test identifies agent with activity against generalized tonic-clonic convulsions, whereas PTZ induced convulsion model identifies agent with activity against myoclonic and generalized seizure by evaluating onset of jerk, straub tail, onset of clonus and extensor parameters [24]. The bioactive isolated compound from BMME was confirmed as lupeol by spectral data analysis and possessed anticonvulsant effect in PTZ induced convulsions at 20 mg/ kg p.o. may be through GABAA mediated action. This indicated that the activity of BMME against convulsion was because of triterpine (lupeol) present in the stem of Butea monosperma (Lam.) Kuntze. |
Conclusion
|
| In conclusion the result of the present study indicated that the methanol extract of stem of Butea monosperma and its isolated bioactive component may be of benefit in the treatment of convulsion. These findings further provides validity for the use of the plant for the management of convulsion in traditional medicine. Further research is encouraged in the area of determining the mechanism of action of the isolated compound lupeol by biogenic amine levels in mice brain responsible for the antiepileptic activity. |
Tables at a glance
|
 |
 |
 |
 |
 |
| Table 1 |
Table 2 |
Table 3 |
Table 4 |
Table 5 |
|
Figures at a glance
|
 |
 |
 |
 |
 |
| Figure 1 |
Figure 2 |
Figure 3 |
Figure 4 |
Figure 5 |
|











