Key words
Borna Disease Virus (BDV), BDV p40, BDV p24, equines, Real Time PCR, Costa Rica.
Introduction
Borna Disease was described more than 200 years ago in South Germany as a fatal neurological disorder in equines and ovines [1,2].
Borna Disease Virus (BDV) is associated with abnormalities in the development of brain structures [3, 4]. Behavioral studies in animals infected with BDV suggest that some neurologic disorders could be associated with this virus [2, 5].
BDV is a member of the Mononegavirales, Bornaviridae family. It is a non-cytolytic, non-segmented, enveloped virus with a negative-sense RNA genome [6]. It has a spherical morphology 70 to 130 nm in diameter, with a dense internal core (50-60 nm) and an external membrane with 7 nm long spicules [7-10].
The BDV genome (~8.9 kb) is typical of the Mononegavirales. It has six open reading frames (ORF) that code for several polypeptides: p40, p24, p10, p16, p56 and p180. p40 corresponds to the viral nucleoprotein (N) and p24 to the phosphoprotein (P) [9,11, 12] Gene X codes for a nonstructural protein that interacts with the P cofactor of the viral polymerase, it inhibits apoptosis and promotes viral persistence [11, 13-15].
Four strains of BDV have been described: Borna V, Borna HE/80, Borna No/98 and Borna H1766. Strain H1766 has 95-98% homology with strains V and He/80, and only 81% with Borna No/98. Other minor variants have been reported as avian bornavirus [1, 16-18].
In Central Europe, neurological disorders have been observed in horses, sheep and other farm animals infected with BDV. The neuronal cells of several species can be infected in vitro with BDV; however, rats are the most susceptible animal. Neurological symptoms are the result of the immune response against persistently infected CNS cells [6, 19, 20].
It is tempting to speculate that BDV might be linked to some psychiatric disease syndromes such as affective disorders (depression, psychosis, schizophrenia) and to idiopathic acute or chronic encephalitis. In the 1980s, the first significant serological evidence for BDV human infection was reported. However, despite more than two decades of study reporting serological, pathological and virological evidence of BDV infection in humans, medical and scientific acceptance of the human as a natural target of BDV is still uncertain [2, 5].
Several methods have been used for detection of BDV infection: indirect immunofluorescence (IIF), Enzyme-Linked Immunosorbent Assay (ELISA), post mortem immunohistochemistry,Flow Cytometry, in situ hybridization, RT-PCR, and nested RT-PCR. However, diagnosis is still difficult [2, 5, 16, 21].
Primary neuronal cell cultures (rat C6 glioma and human oligodendroglial cells) are used for viral isolation. Animal models like rats and rabbits are also used for in vivo infections [5, 14, 22].
Real Time RT-PCR allows the continuous quantification of PCR products by using labeled oligonucleotides [23]. It is specific, rapid, reproducible and very sensitive. It allows detection of very low genome copy numbers.
Due to few circulating particles, BDV is very difficult to detect in blood samples. It is estimated that approximately 1 cell is infected in 105-106 mononuclear cells. Real Time PCR has enough sensitivity to detect this virus [5, 21].
We developed a Real Time PCR for BDV detection, it is based on main antigens p40 and p24. It was applied for the first time in Costa Rica in equine and human samples.
Methods
Samples
Anticoagulated blood samples from 120 healthy horses from different regions of Costa Rica (Guápiles, Tilarán, Miravalles, Santa Cruz, Coronado, Santa Ana, Moravia, Liberia, San Carlos, Heredia) and 76 human samples (51 bipolar patients and 25 from apparently healthy volunteers) were collected with ACD vacuum tubes.
Extraction of cellular and viral RNA from leukocytes
Leukocytes were isolated by centrifugation in a Ficoll® cushion for 30 min at 900 g. The leukocyte layer was extracted, washed and suspended in PBS.
A QIAamp® RNA Blood Mini Kit (QIAGEN) was used for extraction according to the procedure recommended by the fabricant.
Reverse transcription (RT)
A RevertAid H Minus First Strand cDNA Synthesis Kit (Fermentas ®) was used for reverse transcription according to the procedure recommended by the fabricant.
Plasmid extraction
The plasmid pGEX-5x-3 was kindly provided by Dr. Fernando Puerto, University of Yucatán, Mexico. The construct (inserts for p24 and p40) was maintained in the Escherichia coli strain BL21. Cultures were grown in LB (Luria-Bertoni) Agar Amp (50 mg/ml ampicillin, Fermentas®) and then in liquid LB (LB Liq Amp Fermentas®).
Plasmid DNA was extracted by alkaline lysis [24] and the concentration was estimated by fluorometry.
Real Time PCR
A final point TaqMan Real Time PCR (Plus Minus Assay) was used to detect BDV RNA in blood samples. Primers and probes were designed for p24 and p40, based on consensus sequences for Borna V, Borna HE/80, and Borna H1766. Probes and primers for Borna No98 were designed separately because this strain has only 80% homology with the others. (Table 1).
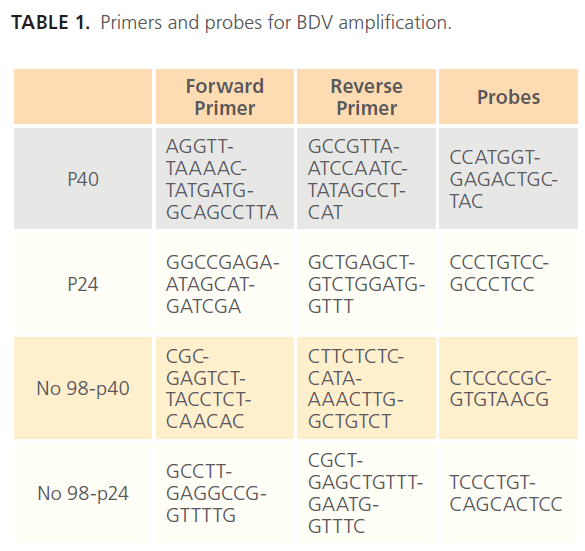
Table 1: Primers and probes for BDV amplification.
The probes were labeled at the 5’ end with a FAM (6-carboxyfluorescein) reporter. Primers and probes were produced by Applied Biosystems®.
PCR Amplification
A Real Time Assay was run in an ABI 7500 PCR System, (Applied Biosystem®).
For each amplification reaction, 10 ml of TaqMan Universal ® PCR Master Mix, 1 ml of 20X assay mix (probe and primers) and 9 ml of cDNA diluted in RNase free water were mixed. Conditions for PCR were: 10 minutes at 95ºC to activate the polymerase, followed by 40 cycles, 15 seconds denaturing at 95ºC and extension at 60ºC for 1 minute. The sensitivity of the assay was determined using 107 to 101 molecules per reaction of p40 and p24.
A BLAST alignment with the GenBank Database (www.ncbi. nlm.nih.gov) was run to determine the theoretical specificity of primers and probes.
Results
Through an estimation of the number of plasmid copies, we determined that the dynamic range for p24 was 102. After the Ct 40 the results were not reliable.
Similar results were obtained for p40, with a dynamic range of 103. In the case of Borna No-98 p24 and p40, a standard curve was not carried out because this sequence is different from the other virus strains and from the p24 and p40 inserts in the plasmids.
The specificity of the reaction was determined through BLAST alignment of the real time PCR products with nucleotide sequences from the GenBank (www.ncbi.nlm.nih.gov). This revealed that these sequences are unique for BDV.
We detected five bipolar human samples positive for p24 (9.8%), three positive for p40 (5.8%), and one positive for strain N98 p40, with a total of 17.6% positive human samples. All healthy volunteers were negative (table 2).
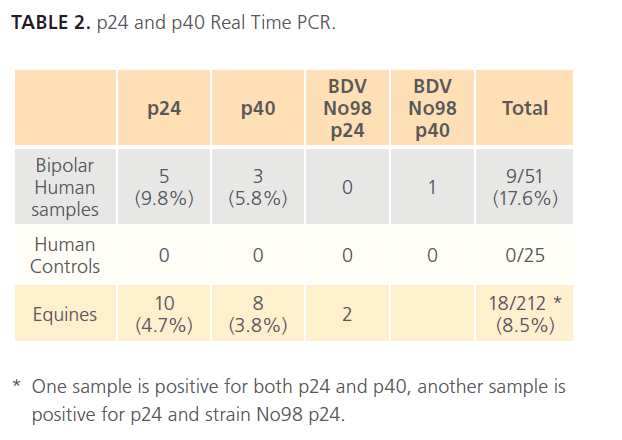
Table 2: p24 and p40 Real Time PCR.
Ten of the equine samples were positive for p24 (4.7%), eight were positive for p40 (3.8%), and two were positive for strain No98 p24 (0.9%), with a total of 8.5% positive equines samples (Table 2).
The PCR products were sequenced on an ABI 3130 (Applied Biosystem). The Real Time PCR products were expected to be approximately 120 bp for p24 and 105 bp for p40. After sequencing we obtained products between 61 to 80 bp, which were aligned (BLAST) in the Gen Bank (www. ncbi.nlm.nih.gov) and similarity was only seen with BDV (93 to 100% similarity). However, the size of the sequences is small, automated systems usually lose 50 to 100 nucleotides at both ends, and sequence reagents can not be altered to improve efficiency.
Discussion
BDV infection yields low viremia and attacks mainly the central nervous system. There are several methods described to detect BDV, but sensitivity is a key factor. Serology has low sensitivity due to low levels of BDV antibodies; therefore, molecular methods are being developed in several laboratories worldwide.
This report describes the standardization of a Real Time RTPCR to detect BDV.
When used with dilutions of the plasmids with the p24 and p40 inserts, the technique detected as few as 100 copies; however, the technique is able to detect fewer molecules.
Interestingly, differences were observed in the relationship between p24 and p40, which varied according to the infection course. Schneider and collaborators report in the acute phase, p40 levels were higher, while in persistent infections p24 levels were increased. [25, 26].
Virus replication occurs when the antigen p40 expression is 30 fold higher than antigen p24. The transcription ratio does not affect polymerase activity. Several studies suggest that the p24: p40 ratio is critical for the regulation of viral replication [10, 25]. These results are in agreement with the observation that most positive horses express p24 in correlation with persistent infections [25].
BDV RNA was detected in 8.5 % of the healthy horses in this study. This appears to be a low percentage compared with reports from Europe. Berg et al in 1999 reported a seroprevalence of 24.5% in healthy horses and 57.7% in horses with neurological signs; however, serology detects previous viral infections and PCR detects only acute infections [27].
Most of the positive horses in this study were from Guápiles County, where bovine, equine and swine farms are common. This fact suggests that infection is endemic and that the virus was probably transmitted from horse to horse.
Animals positive for BDV have been detected in many countries, especially in Europe. On the other hand, in Australia, Kamhieh et al in 2006 could not detect the virus in horses [28]. In humans, BDV RNA was detected in 17.6% of the Bipolar I patients. Similar results were observed in Yucatán with positive results in 21.43% of the samples from schizophrenic patients [29] and in Brazil with 33.33% of the samples from psychiatric patients positive for BDV [30]. It is important to note that the samples from apparently healthy volunteers were all negative for BDV.
Positive bipolar patients ranged in age from 30 to 75 years and had been hospitalized several times, suggesting a relationship between the virus and the persistence of symptoms, or the possibility of virus transmission within the hospital. This findings could have important implications in epidemiology and etiology of neuropsychiatric disorders. In Latin America studies on BDV are scarce. This is the first report of BDV in humans and equines in the Central American region.
161
References
- Kolodziejek J, Durwald R, Herzog S, Ehrensperger F, Lussy H, &Nowotny N (2005) Genetic clustering of Borna disease virus natural animal isolates, laboratory and vaccine strains strongly reflects their regional geographical origin. J Gen Virol 86: 385-398.
- Chalmers R, Thomas D & Salmon R (2005) Borna disease virus and the evidence for human pathogenicity: a systematic review. Q J Med 98: 255-274.
- Bode L, & Ludwig H (2003) Borna disease virus infection, a human mental-health risk. ClinMicrobiol Rev 16: 534-545.
- Ovanesov M, Sauder C, Rubin S, Richt J, Nath A, Carbone K &Pletnikov M (2006) Activation of Microglia by Borna Disease Virus Infection: In Vitro Study. J Virol 80: 12141-12148.
- Carbone K. Borna disease virus and human disease (2001) ClinMicrobiol Rev 14: 513-527.
- Staeheli P, Sauder C, Hausmann J, Ehrensperger F, &Schwemmle M (2002) Epidemiology of Borna disease virus. J Gen Virol 81: 2123- 2135.
- Bode L, Reckwald P, Severus W, Stoyloff R, Ferszt R, Dietrich D, et al (2001) Borna disease virus-specific circulating immune complexes, antigenemia, and free antibodies-the key marker triplet determinating infection and prevailing in severe mood disorders. MolPsyquiatr 6:481-491.
- De la Torre J (2002) Bornavirus and the brain. J Infect Dis 186: S241-S247.
- Hornig M, Briese T, &Lipkin W (2003) Borna disease virus. J Neurovirol 9:259-273.
- Pérez M, Sánchez A, Cubitt B, Rosario D & de la Torre J (2003) A reverse genetics system for Borna disease virus. J Gen Virol 84:3099- 3104.
- Tomonaga K, Kobayashi T, & Ikuta K (2002) Mollecular and celular biology of Borna disease virus infection. Microbes and Infection 4: 491-500.
- Kraus I, Eickmann M, Kiermayer S, Scheffczif H, Fluess M, Richt J, et al (2001) Open reading frame III of Borna disease virus encodes a nonglycosylated matrix protein. J Virol 75: 12098-12104.
- Poenisch M, Wille S, Ackermann A, Staeheli P &Scheneider U (2007) The X Portein of Borna Disease Virus Serves Essencial Functions in the Viral Multiplication Cycle. J Virol 81(13): 7297-7299.
- Poenisch M, Burger N, Staheli P, Bauer G & Schneider U (2009) Protein X of Borna Disease Virus Inhibits Apoptosis and Pomotes Viral Persitence in the Central Nervous Systems of Newborn-Infected Rats. J Virol 83: 4297-4307.
- Chmid S, Mayer D, Schneider U, Schwemmle M (2007) Functional Characterization of the Major and Minor Phosphorylation Sites of the P Protein of Borna Disease Virus. J Virol 81: 5497-5507.
- Nowotny N, Kolodziejek J, Jehle C, Suchy A, Staeheli P, &Schwemmle M (2000) Isolation and characterization of a new subtype of Borna disease virus. J Virol 74: 56-55-5658.
- Staeheli P, Rinker M &Kaspers B (2010) Avian Bornavirus Associated with Fatal Disease in Psittacine Birds. Minireview.J Virol 84: 6269- 6275.
- Clemente R, Sisman E, Aza-Blanc P & Torre JC (2010) Identification of Host Factors Involved in Borna Disease Virus Cell Entry through a Small Interfering RNA Functional Genetic Screen. J Virol 84: 3562-3575.
- Ackermann A, Guelzow T, Staheli P, Schneider U &Heimrich B (2010) Visualizing Viral Dissemination in the Mouse Nervous System, Using a Green Fluorescent Protein-Expressing Borna Disease Virus Vector. J Virol 84: 5438-3442.
- Staheli P &Lieb K (2001) Bornavirus and psychiatric disorders-fact or fiction? J Med Microbiol 50: 579-581.
- Cotto E, Neau D, Cransac-Neau M, Auriacombe M, Pellegrin J, Ragnaud J, et al (2003) Borna disease Virus RNA in Inmunocompromised Patients in Sothwestern France. J ClinMicrobiol 41: 5577-5581.
- Nakamura Y, Takahashi H, Shoya Y, Nakaya T, Watanabe M, Tomonaga K, et al (2000) Isolation of Borna disease virus from human brain tissue. J Virol 74: 4601-4611.
- Radonic A, Thulke S, Mackay I, Landt O, Siegert W, &Nitsche A (2004) Guideline to reference gene selection for quantitative real-time PCR. BiochemBiophys Res Commun 13: 856-862.
- Sambrook J, Fritsch E, &Manitis T (1989) Extraction and Purification of Plasmid DNA. In: Molecular Cloning, 2nd ed. Cold Spring Harbor Laboratory Press USA. pp. 1.25-1.28.
- Schneider U, Naegele M, Staeheli P &Schwemmele M (2003) Active Borna Disease Virus Polymerase Complex Requires a Distinct Nucleoprotein-to-Phosphoprotein Ratio but No Viral X Protein. J Virol 77: 11781-11789.
- Geib T, Sauder C, Venturelli S, Hassler C &Staeheli P (2003) Selective Virus Resistance Conferred by Expression of Borna Disease Virus Nucleocapsid Components. J Virol 77: 4283-4290.
- Berg A, Dorries R & Berg M (1999) Borna disease virus infection in racing horses with behavioral and movement disorders. Arch Virol 144: 547-559.
- Kamhieh S, Hodson J, Bode L, Ludwing H, Ward C & Flower R (2006) No evidence of endemic Borna disease virus infection in Australianhorses in contrast with endemic infection in others continents. Arch Virol 151: 709-719.
- Puerto F, Zavala J, Rosado A, &Gamboa J (2004) Serological survey of Borna disease virus in schizophrenec patients from Yucatán, Mexico. Rev Biomed 15:141-147.
- Miranda H, Vargas S, Sanches E, Zusart S, Nakagawa E, &Ehara MA (2006) Detection Of Borna disease virus p24 RNA in peripheral blood cells from Bazilian mood and psychotic disorder patients. J Affect Dis 90:43-47







