Background: Duchenne and Becker muscular dystrophy (DMD/BMD) are X-linked recessive disorders caused by mutation in dystrophin gene. Patients with muscle weakness came to hospital for improvement their child. Physician advice to dystrophin gene analysis who have high Cpk (Creatine kinase) value and muscle weakness.
Methods: We collected 61 DMD/BMD patient specimens for genetic analysis in which three females were relatives. Multiplex polymerase chain reaction was done for detecting deletion of dystrophin gene.
Results: The overall mutation detection rate was 72.4% (21/29) in DMD/BMD patients, identifying deletions in 58.6% (17/29). Most of the deletions were confined to the central hot spot region between exons 44 and 55 (52.9%, 7/19).
Conclusions: In this study we found that the Exons 50, 51, 48 and 52 are most frequently deleted. The frequency of deletions in exon 50 (21.31%) was the most common deletion frequently associated with our Bangladeshi sample males. This study will help to set up an effective platform for prenatal diagnosis in Bangladesh.
Keywords
Duchene muscular dystrophy (DMD), Xlinked, Mutation, Prenatal diagnosis.
Introduction
Duchene muscular dystrophy and Becker muscular dystrophy are allelic X-linked recessive disorders that are caused by mutation of the DMD gene that located at Xp21 [1]. It is the most common in childhood muscle disease in children and the proportional value of 1:3500 lives male births [2]. Both DMD and BMD are caused by mutations in the Dystrophin gene, which is the largest human gene, spanning 42,200 kb on the X chromosome and occupying roughly 0.1% of the genome [3]. The DMD gene contains 79 exons encoding a 14,000 bp messenger RNA transcript that is translated into the protein Dystrophin. The Dystrophin is localized on the cytoplasmic surface of the plasma membrane of skeletal and cardiac muscle cells [4]. Dystrophin in the sarcolemma of muscle fibers, is almost absent in DMD and decreased in BMD [5]. Disruption of the blood-brain barrier has been seen to be a noted feature in the development of DMD [6]. Random inactivation of one of the two X chromosomes occurs during early embryonic development, leaving active 50% of the maternally derived chromosomes and 50% of the paternally derived ones [7]. Non-random X-chromosome inactivation induces fewer than 50% of the nuclei which may have the normal dystrophic gene, resulting in clinical manifesting female carriers [8]. Majority of large deletions initiate at the 5’ region of the dystrophic gene, for example: large deletion of introns 2 to 42. Increased breakpoint frequencies near the 5’ end are largely due to large sizes of some introns [9]. Patients with deletions in the amino terminal domain I typically had low protein levels and are very severely affected irrespective of disruption or maintenance of the reading frame, thereby suggesting this domain is functionally critical part of the dystrophic, while loss of just the carboxyl terminus often caused BMD [10]. Previous reports suggest that large deletions account for approximately 65% of DMD mutations and 35% of BMD mutations [4]. Studies of Duchene muscular dystrophy (DMD) prevalence suggest estimation of 0.1% in South Africa, 10.5 to 1.0 % in Asian countries, 0.7% to 1.0 % in North America and 0.2% to 2.8 % in European countries [11].
Most of the patients with DMD related lived in rural area and below poverty line and have no data record of this disease in Bangladesh. The current methodologies used for detecting mutations in the dystrophic gene including multiplex PCR, Southern blotting, multiplex ligation-dependent probe amplification [12-14].This study is first time analysis common DMD/ BMD gene analysis in Bangladeshi population. Firstly, patients came to hospital with their child with different types of problem facing such as most of the child are not capable to stand up, no properly sit down, delay growth development, muscle pain and so on. Genetic testing is always necessary even if DMD is first confirmed by high Cpk value. In this study we screened majority of the deletions (81.2 %) were located at distal hot spot region that encompasses exons 42-53 and 9.5% of the deletions were located at the proximal hot spot region (exons 2-19). Exons 48, 50, 51 and 52 are most frequently deleted. The frequency of deletions in exon 50 (21.31%) was the most common deletion frequently associated with our Bangladeshi sample males.
Materials and Method
Samples preparation
61 blood samples were collected from the patients with high cpk value or muscle biopsy that indicated to problem of DMD/ BMD. The patients who came to Dhaka Shishu Hospitals for treatment were selected as subjects. Written informed consent was taken from all participants to make the study ethically sound (Figure 1 and 2). The Peripheral blood was collected into EDTA containing tube. Blood samples from patients were stored in 4°C. DNA was isolated from peripheral blood using PureLink@ Genomic DNA Purification Mini Kit (Genera, USA). This method relies on phase separation by centrifugation of a mix of the aqueous sample. The kit was used according to the manufacturer’s instructions. Extracted DNAs were tested to determine the purity and concentration of DNA per micro-litter volume through Qubbit 2.0 Fluor meter. This extracted DNA samples stored in -200 C for long time (Table 1 and 2).
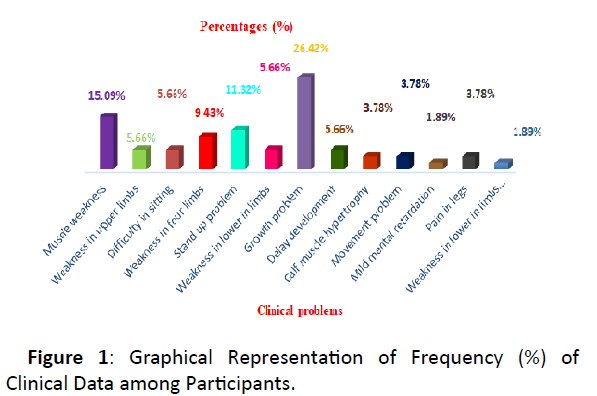
Figure 1: Graphical Representation of Frequency (%) of Clinical Data among Participants.
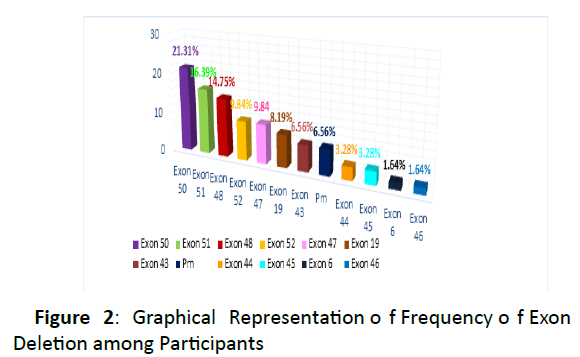
Figure 2: Graphical Representation of Frequency of Exon Deletion among Participants
| Clinical Symptoms |
Frequency (n) |
Percentage (%) |
| Muscle weakness of the pelvic area/ thighs/ shoulders/ calves |
8 |
15.09% |
| Weakness in upper limbs |
3 |
5.66% |
| Difficulty in sitting |
3 |
5.66% |
| Weakness in four limbs |
5 |
9.43% |
| Stand up problem |
6 |
11.32% |
| Weakness in lower in limbs |
3 |
5.66% |
| Growth problem |
14 |
26.42% |
| Delay development |
3 |
5.66% |
| Calf muscle hypertrophy |
2 |
3.78% |
| Movement problem |
2 |
3.78% |
| Mild mental retardation |
1 |
1.89% |
| Pain in legs |
2 |
3.78% |
| Weakness in lower in limbs & difficulty in walking |
1 |
1.89% |
Table-1: Frequency (%) of Clinical Data among Responders.
| Deletional exon name |
Frequency (%) |
| 50 |
21.31 |
| 51 |
16.39 |
| 48 |
14.75 |
| 52 |
9.84 |
| 47 |
9.84 |
| 19 |
8.19 |
| 43 |
6.56 |
| Pm |
6.56 |
| 44 |
3.28 |
| 45 |
3.28 |
| 6 |
1.64 |
| 46 |
1.64 |
Table 2: Frequency of Exon Deletion among Study Subjects
Identification of mutational hotspot
Mutation mostly deletion was analyzed by four multiplex PCR sets containing 18 pairs of primers. These primers were divided into four groups. First group was consisted of 4 (196), 48 (506), 50 (271), 51 (388), 52 (113) exon deletion primer, second reaction set was 43 (357), 45 (547), 47 (181), 53 (212), third group was consisted by 3 (410), 8 (360), 13 (238), 42 (195), Pm (535), fourth group included 6 (202), 19 ((459), 44 (268), 60 (139).
10μl of the pcr product are loaded on 2% agarose gel TAE buffer with Ethidium bromide solution and run at 100 volt and 300 at for 30 min. In addition, bands are visualized on UV transiluminator and then photographed by using photo documentation system. All samples were analyzed with positive and negative controls for particular mutation. An 861bp internal control band was also amplified in all reactions indicating successful PCR.
Results
Muscle weakness (15.09%), stand up problem (11.32%), growth problem (26.42%) were commonly experienced clinical problem among study subjects where growth problem (26.42%) had been found to be most prevalent.
Exon 50 deletion and exon 51 deletions were most commonly found and the frequency was 21.31% and 16.39%. On the other hand exon 6 deletions (1.64%) & exon 46 deletion (1.64%) were less common among study subjects.
Discussion
There is no dystrophic gene deletion data available among Bangladeshi individuals. Although in American, deletion of exon 45 is most common [15]. In Thai population, Most extensive deletions ocnsisted ofe xon 14 deletions. Most ferquently deleted exons were exon 44-52 [16-18]. In Pakistan, most frequently deleted exons (frequency wise) were 50 (15.11%), 6 (12.79%), 47 (10.46%), 13 (8.13%) and 52 (4.65%) with deletion frequencies [19]. Chinese DMD exon deletions in local Chinese patients were significantly lower (34.3%) than the commonly quoted 60%. This indicated an ethnic or regional difference in predisposition to DMD exon deletions [20].
In Asian Central region is the deletion hotspot in the following 3 Asian populations: Singaporeans (61.9%); Japanese (70.5%); Vietnamese (72.7%) [21]. In Western India, maximum deletions initiated at exon 45 (76.1% of the cases) [22]. In Eastern (parts of West Bengal & a few eastern states) ~79% deletions were found in the central and 17.91% at the proximal (5’) hot spot region [23]. In Southern part of India, deletional hotspots were found in exons 45, 47, 49 and 50 [24].
Our study was undertaken for first time in Bangladesh to detect the DMD/BMD exon deletional mutations in Bangladeshi population.
The study found that, exon 50 deletion and exon 51 deletion were most common DMD mutation in Bangladeshi patients where the frequency were 21.31% and 16.39% respectively. The third common deletional mutation was exon 48 (14.78%). The deletion frequency of both exon 52 & exon 47 deletion were 9.84%. Exon 19 deletion exon 43 deletions were was found in 8.19% & 6.56% of cases respectively. The frequency of both exon 44 deletion & exon 45 deletion were 3.28% On the other hand exon 6 deletion (1.64%) & exon 46 deletion (1.64%) were less common in our study subjects.
The main symptom of DMD was muscle weakness associated especially those of the hips, pelvic area, thighs, shoulders, and calves in study subject. Muscle weakness also occurs later, in the arms, neck, and other areas. Calves are often enlarged.DMD/BMD patients in our study experienced various types of clinical problem. Muscle weakness (15.09%), stand up problem (11.32%), growth problem (26.42%) were commonly experienced clinical problem among study subjects where growth problem (26.42%) had been found to be most prevalent. In this study the majority of exon mutation occur in exon 50 and their physiological effect for the deletion of exon 50 were identified they were – muscle weakness , weakness in upper limbs , stand up problem, difficult in sitting. The mutation of exon 51 and 48 was also frequent occurred disorder , their frequency were about 16.39% and 14.75%.The physiological effect for the deletion of exon 51 and 48 were identified they were – muscle weakness, mild mental retardation , weakness in limbs , pain in legs and weakness in four limbs. The deletion of exon 48, 52, 47 , 19 , 43 were mild frequent occurring disorder and their physiological defect were - Movement Problem , Calf muscle hypertrophy , weakness in four limbs , Difficult in sitting and so on. The deletions of exon 44, 45, 6, 46 were identified as less frequent occurred disorder but they present in a numbers of population. From this study we concluded that the frequency of deletion in exon 50 (21.31%) was the most common deletion frequently associated with our Bangladeshi sample males.
Conclusion
This study provides the pattern of dystrophic gene deletion from Bangladeshi population which responsible for various kinds of muscle disorder , this study will be helpful in molecular diagnostics for prenatal diagnosis and prevention of genetic disorder by proper counseling in different areas of Bangladesh. I think that MPLA method is rather best method than Multiplex PCR method to analysis Dystrophin gene
39891
References
- Muntoni F, Torelli S, Ferlini A (2003) Dystrophin and mutations: one gene, several proteins, multiple phenotypes. Lancet Neurol 2:731–740.
- Emery AE (1991) Population frequencies of inherited neuromuscular diseases: a world survey. NeuromusculDisord. 1:19–29.
- Prior TW, Bridgeman SJ. Experience and strategy for the molecular testing of Duchenne muscular dystrophy. J MolDiagn. 2005;7:317–326.
- Hoffman EP, Brown RH, Jr, Kunkel LM. Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell. 1987;51:919–928.
- Roberts RG, Bobrow M, Bentley DR. Point mutations in the dystrophin gene. ProcNatlAcadSci U S A. 1992;89:2331–2335.
- Zhao Z, Nelson AR, Betsholtz C, Zlokovic BV. Establishment and Dysfunction of the Blood-Brain Barrier. Cell. 2015;163(5):1064-1078.
- Traverso M, Malnati M, Minetti C, Regis S, Tedeschi S, et al. (2006) Multiplex real-time PCR for detection of deletions and duplications in dystrophin gene. BiochemBiophys Res Commun 339:145–150.
- Yau SC, Bobrow M, Mathew CG, Abbs SJ. Accurate diagnosis of carriers of deletions and duplications in Duchenne/Becker muscular dystrophy by fluorescent dosage analysis. J Med Genet. 1996;33:550–558.
- Gatta V, Scarciolla O, Gaspari AR, Palka C, De Angelis MV, et al. (2005) Identification of deletions and duplications of the DMD gene in affected males and carrier females by multiple ligation probe amplification (MLPA) Hum Genet. 2005;117:92–98.
- Lalic T, Vossen RH, Coffa J, Schouten JP, Guc-Scekic M, et al. (2005) Radivojevic D, Djurisic M, Breuning MH, White SJ, den Dunnen JT. Deletion and duplication screening in the DMD gene using MLPA. Eur J Hum Genet 13:1231–1234.
- Paul A. Romitti, Yong Zhu, Soman Puzhankara (2015) Prevalence of Duchenne and Becker Muscular Dystrophies in the United States. Pediatrics 135: 513-21.
- Verma PK, Dalal A, Mittal B, Phadke SR. Utility of MLPA in mutation analysis and carrier detection for Duchenne muscular dystrophy. Indian J Hum Genet. 2012;18(1):91-94. doi:10.4103/0971-6866.96667
- Nouri N, Fazel-Najafabadi E, Salehi M, et al. Evaluation of multiplex ligation-dependent probe amplification analysis versus multiplex polymerase chain reaction assays in the detection of dystrophin gene rearrangements in an Iranian population subset. Adv Biomed Res. 2014;3:72. Published 2014 Jan 27.
- Janssen, B., Hartmann, C., Scholz, V. et al. MLPA analysis for the detection of deletions, duplications and complex rearrangements in the dystrophin gene: potential and pitfalls. Neurogenetics 6, 29–35 (2005).
- Barzegar M, Habibi P, Bonyady M, Topchizadeh V, Shiva S. Exon deletion pattern in duchene muscular dystrophy in north west of iran. Iran J Child Neurol. 2015;9(1):42-48.
- Sura T, Eu-ahsunthornwattana J, Pingsuthiwong S. et al. Sensitivity and frequencies of dystrophin gene mutations in Thai DMD/BMD patients as detected by multiplex PCR. Dis Markers. 2008; 25(2):115-21.
- Basumatary LJ, Das M, Goswami M, Kayal AK. Deletion pattern in the dystrophin gene in Duchenne muscular dystrophy patients in northeast India. J Neurosci Rural Pract. 2013;4(2):227-229.
- Thakur N, Abeysekera G, Wanigasinghe J, Dissanayake VH. The spectrum of deletions and duplications in the dystrophin (DMD) gene in a cohort of patients with Duchenne muscular dystrophy in Sri Lanka. Neurol India 2019;67:714-5
- Hassan M J, Mahmood S, Ali G, et al. Intragenic deletions in the dystrophin gene in 211 Pakistani Duchenne muscular dystrophy patients. Pediatr Int. 2008; 50:162-6.
- Lo IF, Lai KK, Tong TM, Lam ST. A different spectrum of DMD gene mutations in local Chinese patients with Duchenne/Becker muscular dystrophy. Chin Med J (Engl). 2006;119(13):1079-1087.
- Lai PS, Takeshima Y, Adachi K; Comparative study on deletions of the dystrophin gene in three Asian populations. J Hum Genet. 2002. 47:552-555.
- Dastur RS, Gaitonde PS, Kaldikar SV. et al. Becker muscular dystrophy in Indian patients: Analysis of dystrophin gene deletion patterns. Neurol India .2008; 56:374-77.
- Basak J, Dasgupta UB, Banarjee TK. et al. Analysis of dystrophin gene deletions by multiplex PCR in eastern India. Neurol India. 2006; 54:310-1.
- Swaminathan B, Shubha GN, Shubha D. et al. Duchenne muscular dystrophy: a clinical, histopathological and genetic study at a neurology tertiary care center in Southern India. Neurol India 2009; 57(6):734-8.







