Keywords
Scars; Scar visibility; Cicatrice visibility; Wound-dressings
Case Report
In order to minimize the cicatrice visibility by off loading the surgical incision, a new device has been developed by the two above mentioned researchers, consisting in a pair of dressings adhering to the skin, right and left of the incision, trhough pressure-sensitive adhesives. These dressings are made of a composition based on silicone elastomers embedding small magnets, so that the left element continuously attracts the right one. This mutual attraction is to be considered as permanent, even weeks or months after applying [1,2].
A first clinical trial has been performed with positive results as summarily described hereunder. After a first trial on animal (Université de Liège, October 30, 2015, with mention, (the magnetic device allowed a better cicatrization compared to the one with staples) the clinical trial on human received the approval of Ethical Committee of Erasme Hospital, Université Libre de Bruxelles, on January 7, 2016, as well as the authorization for clinical investigation by Federal Agency for Medicines and Medical Devices, Brussels [1].
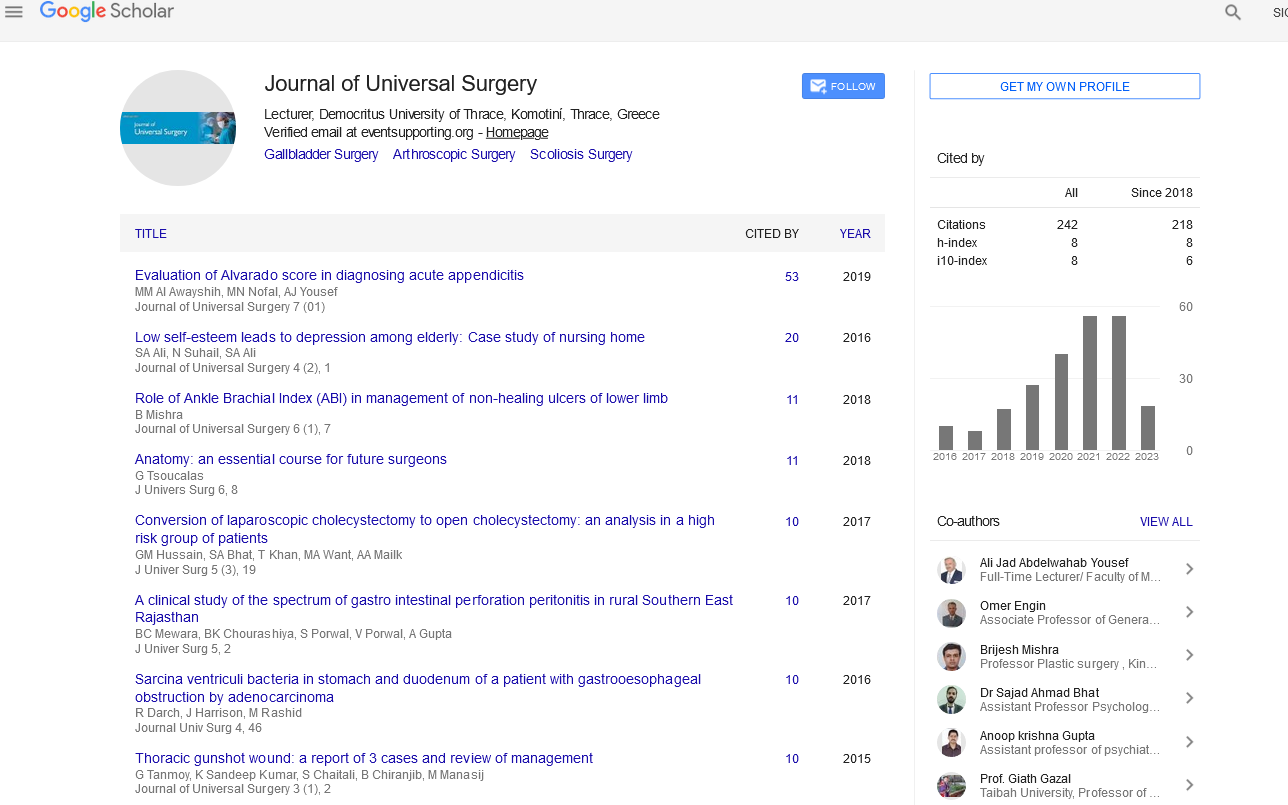
The first picture here shows the existing scar, length 105 mm, in the back of the patient (male with white complexion) dated from several years and widened (Figure 1).
Figure 1: The scar in the back of the patient (male with white complexion), dated several years, and widened.
The original intervention on this patient has been lombar hernia discal surgery. After statement that the scar had notably widened under skin traction, the patient has required a first time, an excision of it, and re-suturing. The exact date of it had not been noted by the patient, - it was probably about 5 years ago, at least [3]. Then, appeared a widening again, as visible here.
On January 11, 2016, the cicatrice has been again excised, on a length of 95 mm, with cold blade and electrocautery, depth 1 cm (Figure 2). The appearance of the curves confirm that a certain effect of «pull» exists by the skin tissues in the back of the patient.

Figure 2: Excision of the cicatrice, length 95 mm, depth 10 mm, with cold blade.
After intradermal suturing, 3 prototypes of the new device magnetic dressings, each 20 mm width, have been placed (Figure 3).

Figure 3: Magnetic dressings placed centered above the cicatrice, after withdrawal of their (key).
Brief description of the novel magnetic dressing
It consists in a pair of adhesive wound-dressings, provided with small magnets, embedded in a matrix of silicone elastomers, so that they attract mutually right and left of the incision. The magnets, neodyme type, are 4 mm rods, gold plated. The device can be made as narrow as desired witha minimum of 9 mm, in order to conform easily to a curved incision. Here the width is 20 mm. The length may preferably not be less than 25 mm each side, in order to benefit of enough skin adhesived surface (this exclude the placing on certain places as the nose, for instance). Each element here is 40 mm long. It is important to note that the magnets are protruding out of the item whre they are embedded, what allows the visibility of the incision and cicatrice at any moment, to monitor for instance any departure of inflammation or the like. Placing the pair of dressings is simple: they are temporarily maintained distant of each other by a flat surface acting as (separation-key) compressed between the magnet ends, so that the whole remains handlable, manipulable. After firm contact-adhesiving of both elements of the pair, the separation-key is withdrawn, what allows the start of a stroke of movement reuniting the two elements of the pair, with tensile effort in the skin. The separation-key is an essential feature of the system (and the patentable one). Adjusting the thickness of it, allows to monitor the amount of elongation for a given effort. In the present case, attraction effort is 8 newtons (800 grams) per element, and the key thickness 4 mm.
The dressings in the present case were allowed to stay 3 months adhesived to the back of the patient. Placing and withdrawing the elements do not require a tool (except of course the «key» which is discarded). They are of cheap cost [3].
Depending from the rate of perspiration of the patient’s skin, the adhesion of the elements may last 5 to 45 days. On the present case they have been changed every week. The patient’s back was seat of serious perspiration. On the arm of same patient, the duration had been 45 days. (Perspiration rate, as known, depends of many factors such insulation, room temperature, season of the year, ingested food, etc).
In the present case the dressings have been withdrawn after 110 days, and the (Figure 4) hereafter, taken. A widening of the scan seems to have been avoided, and an exerted eye will probably guess that only a thin cicatrice will remain visible. Of course the definitive appearance should be the object of examination and photograph after 1 year [4].
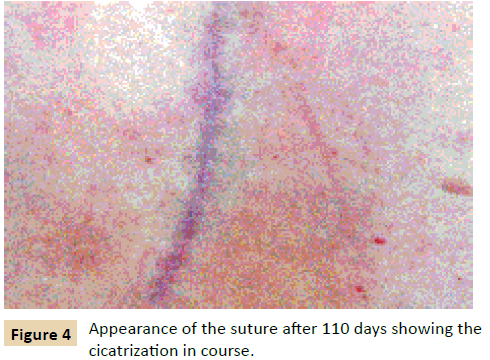
Figure 4: Appearance of the suture after 110 days showing the cicatrization in course.
A first European patent application has been applied for, and a PCT obtained under the number PCT/EP2015/078885. The text can be consulted on the site of European Patent Office.
The patent application is owned by the Belgian company Continental Structures, research office, based in Zaventem (Belgium), sponsor of the clinical trial at Brussels University.
To be noted that an analogous device had been developed under the trademark «Embrace» in the USA as soon as 2010 by Neodyne BioSciences Inc. of Palo Alto, California, and is on the market since (2014). The Belgian new proposed item is claimed to be cheaper, more simple of use, and, important point, allows permanently to see the cicatrice between the two attracting elements.
9349
References
- (2015) Approval of Clinical Investigation on Device for Suturing Skin Wounds based on mutual attraction of the components, protocol P2015/289, by Federal Agency for Medicines and Health Products, 40/40 VHorta place, Brussels, Belgium.
- David L. Dunn Wound Closure Manual,published by Johnson & Johnson Inc. ,Johnson & Johnson Plaza, New Brunswick (New Jersey, USA), describing some mechanical methods for offloading surgical incisions p: 78.
- Lim AF, Weintraub J, Kaplan EN, Januszyk M, Cowley C, et al. (2014) The embrace device significantly decreases scarring following scar revision surgery in a randomized controlled trial.PlastReconstrSurg133: 398-405
- Gurtner, Dauskardt RH, Wong VW, Bhatt KA, Wu K, et al. (2011) Improving cutaneous scar formation by controlling the mechanical environment: large animal and phase I studies. Ann Surg 254: 217-225.