Keywords
Nitrous oxide; Laughing gas; Vitamin B12; Spinal cord; Rehabilitation; Substance related disorders
Abbreviations
CSF: Cerebrospinal Fluid; ICF: International Classification of Function, Disability and Health; IVIG: Intravenous Immunoglobulin; MCV: Mean Corpuscular Volume; MESH: Medical Subject Heading; MMCoAM: Methylmalonyl-Coenzyme- A Mutase; MTHFR: Methylenetetrahydrofolate reductase; MTR: Methyltransferase; N2O: Nitrous Oxide; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; WHO: World Health Organisation
Introduction
Nitrous oxide (N2O) was first utilized in a recreational setting prior to its well documented medical use in the fields of medicine and dentistry for its analgesic and anaesthetic properties [1]. The first safety concerns surrounding the use of N2O in anaesthetics arose following a report published by Lassen, et al. [2], who raised the potential of bone m arrow suppression following N2O anaesthesia. Numerous reports have been published in the medical literature documenting the potential neurological, haematological and cardiovascular sequelae to patients who received the nitrousbased anaesthetic and health workers who were exposed to the residual gas [3].
There has been a steady increase in the recreational use of N2O on a global scale although its regular use has been declining in anaesthetics internationally. The global drug survey of 2017 which reported the recreational drug habits of 5,900 individuals in the UK and 1,20,000 individuals internationally over a 12-month period demonstrated a rise of nitrous oxide use from 20% in 2014 to 31% in 2017. This was a much steeper rise compared to other psychedelics such as Ketamine and LSD [4].
The gain in popularity of N2O in the recreational drug scene is likely because it is legal, low cost of $12 AUD (approx.) for a box of 10 canisters and undetectable on routine drug screens. Even though internationally the sale of Nitrous oxide incurs large penalties to suppliers if sold for recreational use, the gas is still freely available via catering outlets, convenience stores and via various internet platforms.
Nitrous oxide for recreational purposes is commonly sold as prefilled balloons or small pressurized canisters termed “whippets” designed for the food industry used in the preparation of whipped cream. It has a rapid onset of action which lasts for roughly 20 seconds and a rapid offset with many users being able to resume their daily activities within two minutes [1].
There is well documented evidence to suggest that there are several adverse effects associated with the acute and chronic use of nitrous oxide. Nitrous oxide exerts its adverse effects by inactivating vitamin B12 which is key in myelin production and DNA synthesis. This results in numerous clinical manifestations including subacute combined degeneration of the spinal cord, peripheral neuropathy, functional vitamin B12 deficiency and various psychiatric sequelae [3].
Based on the available literature, the neurological deficits associated with nitrous oxide use have far reaching consequences on mobility, activities of daily living, community participation, social engagement and overall quality of life [3,5,6]. This would imply that chronic nitrous oxide users who present to hospital could potentially benefit from a period of rehabilitation prior to discharge. However, this is not well reflected in the available literature with many case reports failing to document if patients were even offered a period of rehabilitation prior to discharge.
The aim of this systematic review is to evaluate published case reports and case series to answer the following key disability focused questions in keeping with the World Health Organisation (WHO) International Classification of Function, Disability and Health (ICF):
1. What are the key neurological impairments in individuals following recreational nitrous oxide abuse?
2. What are the key activity limitations from a mobility and functional perspective associated with recreational nitrous oxide abuse?
3. Does a multidisciplinary rehabilitation program aid recovery to promote improved participation in everyday life situations in this patient group?
Methods
Eligibility and exclusion criteria
Studies were included or excluded based on the following pre-defined study criteria. Table 1 provides a summary of the eligibility criteria for the current review.
| Parameters |
Inclusion Criteria |
Exclusion Criteria |
| Study design |
|
- Animal studies
- Review articles
- Editorials
- RCTs
|
| Language |
- English language
- Non-English language
|
- |
| Age range |
- Adults -19+
- Adolescents – 13-17
|
|
| Gender |
|
- |
| Nitrous oxide exposure |
|
- Anaesthetic exposure
- Analgesic exposure
|
| Clinical Manifestations |
|
- Other clinical manifestations with no associated neurological deficits
- Death
|
Table 1 Inclusion and exclusion criteria.
Data sources
The following databases were searched: PubMed (NLM) (1964- 08.07.2020), MEDLINE (OVID) all including Epub Ahead of Print, In-Process and Other Non-Indexed Citations, Daily and Versions (1946-08.07.2020), Embase (OVID) (1974-08.07.2020), PsychINFO (OVID) (1806-08.07.2020), CINHAL Complete EBSCOhost (1989- 08.07.2020) and Scopus Elsevier (1970-08.07.2020)
Search strategy
The searches were conducted in collaboration with an experienced research librarian as recommended in The Joanna Briggs Institute Reviewers’ Manual [7]. The databases PubMed, Medline, Embase and Scopus were systematically searched to identify literature relevant to the key aims of this review. Additional databases -CINAHL, covering nursing and allied health literature and PsychINFO were also included. Key Medical Subject Heading (MESH) terms were identified including “Nitrous Oxide”, “Laughing Gas”, “Vitamin B12”, “Spinal Cord” and “Substance- Related Disorders”. Through a review of text words in titles and abstracts of the literature found in the databases, new keywords were also identified and included in the final database search. Table 2 provides a list of the specific search queries used in all databases. Hands searching of the bibliographies of selected studies were also performed to identify studies that were potentially not captured in the primary search.
PubMed -NLM
1964-2020 |
"Laughing Gas" OR "Nitrogen Protoxide" OR "Nitrous Oxide"[MeSH Terms] OR "Nitrous Oxide") AND ("Drug Abuse" OR "Drug Addiction" OR "Drug Dependence" OR "Drug Habituation" OR "Drug Use Disorders" OR "Prescription Drug Abuse" OR "Substance Abuse" OR "Substance Addiction" OR "Substance Dependence" OR "Substance Use Disorders" OR "Substance-Related Disorders"[MeSH Terms] OR "Substance-Related Disorders")
Limits (Case Reports, Humans, Adolescent: 13-18 years, Adult: 19+ years) |
Medline OVID
1946-2020 |
*Nitrous Oxide/and exp Substance-Related Disorders/(humans and ("adolescent (13 to 18 years)" or "young adult (19 to 24 years)" or "adult (19 to 44 years)") and case reports) |
PsychINFO
OVID
1806-2020 |
Exp Drug Abuse/or exp Nitric oxide/or exp Inhalant Abuse/or nitrous oxide.mp
and Exp Spinal cord/and Vitamin B12. mp Limit to Human and 200 adolescence <13to 17 years OR 300 adulthood |
CINHAL Complete
1989-2020 |
Inhalant abuse/Substance abuse/Nitrous oxide/Or recreation* “nitrous oxide” ab And Spinal cord/ |
Embase -OVID
1974- 2020 |
Exp human/and exp case report/and *nitrous oxide/and exp abuse/ limits (human and case reports (adolescent <13 to 17 years> or adult <18 to 64 years>) |
Scopus – Elsevier
1970-2020 |
((nitrous and oxide and recreational) OR (illicit and use) AND (spinal and cord)) limit human - ti,ab,kw |
Table 2 Database search strategy.
Study selection
The final search obtained 255 references which were pooled in EndNote X9.3.3 citation manager. A further 5 articles were obtained by hand searching the included articles. 146 duplicates were identified. 150 article abstracts were screened and a further 61 were excluded. 94 full text references were evaluated by 2 independent reviewers. Disagreements between the two reviewers were resolved by consensus. 28 references were excluded. 66 references were included for qualitative synthesis. The formal study selection process is illustrated in the PRISMA Flowchart in Figure 1.
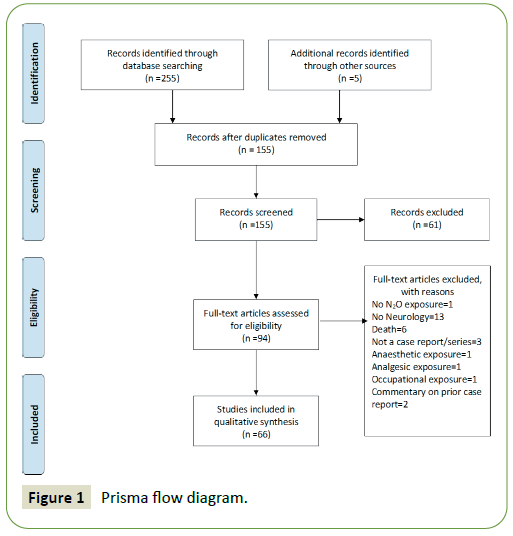
Figure 1: Prisma flow diagram.
Data extraction
The following data were extracted from the eligible articles. Patient characteristics including age, gender, occupation, previous history of drug abuse, presenting neurological impairments, neurological findings on physical examination, laboratory findings, imaging findings, treatments received including inclusion in a rehabilitation program prior to discharge and presence of neurological deficits on discharge. The results were synthesised across studies.
Reporting and risk of bias assessment
The current systematic review was formatted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta- Analyses (PRISMA) statement [8]. The risk of bias assessment was performed by 2 reviewers. We assessed the potential for bias using the Joanna Briggs Institute Critical Appraisal Checklist tool for Case reports and Case series [9]. Disagreements between the reviewers were resolved by consensus.
Results
Study selection and characteristics
The authors screened 155 articles as part of the systematic review. 61 records did not meet the inclusion criteria. 94 full text articles were evaluated and a further 28 articles were excluded. A total of 66 articles were deemed suitable for qualitative analysis. All included studies were case reports or case series. A summary of the individual patient characteristics is available on request.
The 66 articles originated from North America (n=33), Australia (n=8), Taiwan (n=7), United Kingdom (n=6), China (n=5) Netherlands (n=3), Finland (n=1) Canada (n=1), South Korea (n=1) and South Africa (n=1).
Risk of bias assessment
All 66 included articles were subject to critical appraisal using the Joanna Briggs Institute Critical Appraisal Checklist tools for Case Reports and Case Series [9]. All included articles were deemed to have a high degree of bias as outlined in the criteria. There was a high degree of selection and reporting bias as all included articles were case reports and case series. Thus, the results in this study should be interpreted with caution. However, given the lack of high-quality evidence, the available literature can have value to identify trends of clinical sequelae and predictors of recovery post recreational N2O exposure.
Key demographic data of individual studies
66 unique studies were included in this systematic review which included 88 individual case reports. This included 43 males and 32 females. 13 case reports did not mention the gender of the individual patient. The mean age of subjects was 26 years (± 7.51). Only 28 case reports mentioned the individual patient’s occupation which included: university student (n=9), high school student (n=2), medical professional (n=8), professional window cleaner (n=1), carer (n=2), hospital technician (n=1), computer programmer (n=1), writer (n=1), mechanic (n=1), electrical engineer (n=1) and retail worker (n=1). This indicates a higher proportion of users with tertiary qualifications (20/28).
Amount, duration and method of nitrous oxide consumption
The duration of Nitrous oxide exposure was documented in 61 reports, which ranged from 2 weeks to 15 years. The frequency of consumption ranged from daily use to “a few times” per month. The reporting of duration and frequency was very poor with many reports not specifying exact figures. The information provided was also limited with regards to the amounts consumed. Only 57 cases reported quantity consumed which ranged from 0.5 whipped cream cartridges or “whippets” (containing 8 grams of 100% N2O) to 450 per session. The methods of administration were not limited to “whippets” but also included inhaling pure N2O from a tank, anaesthetic machine and a wall outlet in a hospital in one case report.
Apart from nitrous oxide consumption, 43 cases mentioned the use of other substances. The most common included alcohol (58%) and tobacco (42%). 21% denied concurrent use of any other substance. Less commonly reported substances included cocaine (12%), heroin (2%), ketamine (7%), oxycodone (2%), cannabis (7%), ecstasy (2%), benzodiazepines (2%) and amphetamines (5%). Of the 43 cases 42% admitted to poly substance use.
Neurological impairments at presentation and neurological deficits identified on physical examination (Neurological impairments): 78 of 88 cases documented symptoms at presentation. 10 cases which were part of a case serries by Keddie, et al. [10] failed to document the individual patients presenting clinical features. The most common presenting neurological impairments were sensory disturbance which included paraesthesia of the limbs and numbness (72%), gait disturbance (47%) and limb weakness (36%). All presenting neurological impairments are presented in Figure 2.
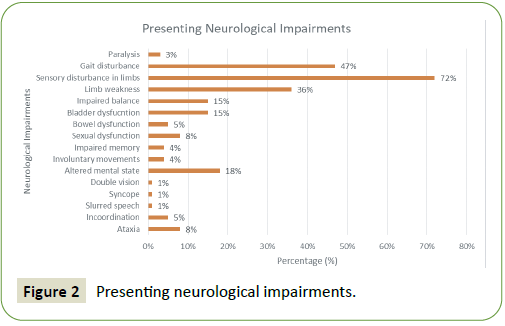
Figure 2: Presenting neurological impairments.
87 of 88 cases had a documented formal neurological examination performed at the time of presentation. One case documented by Lin, et al. [11] did not document a formal neurological examination. The most common findings included loss of vibration sense (68%), proprioceptive loss (59%) and gait ataxia (55%). All documented findings on formal neurological examination are presented in Figure 3.
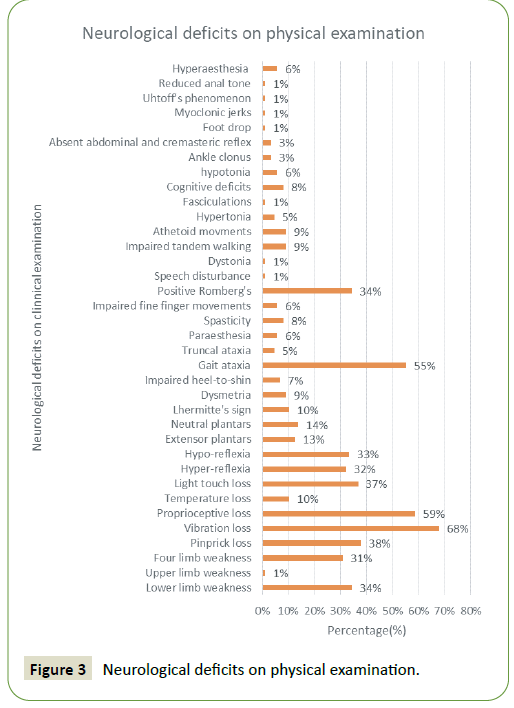
Figure 3: Neurological deficits on physical examination.
The principle diagnoses of the 88 cases included Subacute Combined Degeneration of the spinal cord (n=37), myeloneuropathy (n=15), peripheral neuropathy (n=15), myelopathy (n=14), neurotoxicity from nitrous oxide with no diagnosis specified (n=4), encephalitis (n=1), subacute delirium (n=1) and Methylenetetrahydrofolate reductase (MTHFR) deficiency (n=1).
Laboratory, neurophysiological and imaging findings
2 of the included case reports did not mention formal laboratory results. 30% of the 86 cases had documented anaemia and 70% had no documented anaemia. Interestingly only 4 cases had a past history of anaemia documented. 3 of them had a history of pernicious anaemia and 1 had a history of non-haemolytic anaemia. Of the 29 cases that had information regarding Mean Corpuscular Volume (MCV), 62% of cases had an elevated MCV and 38% of cases had a normal MCV. 29 cases documented an elevated homocysteine level and 30 documented an elevated Methylmalonic acid (MMA) level. 88 case reports had information regarding serum B12, which was low in 65% of cases and normal in 35% of cases. Of note, only four cases with low serum vitamin B12 had a prior history of B12 deficiency documented. Of the 23 cases that had documented Cerebrospinal Fluid (CSF) results, no abnormalities were noted.
Only 39 cases had nerve conduction studies or electromyography performed. Of this, 90% had a positive result and only 10% had a normal result. The most frequent diagnoses included demyelinating polyneuropathy, sensorimotor axonal polyneuropathy and motor axonal neuropathy. One case reported a diagnosis of cervical myelopathy.
87% of the 61 cases where MRI spine results were available had an abnormal MRI result and only 13% had a normal MRI result. The MRI spine results were consistent with findings associated with subacute combined degeneration of the cord which is increased T2 signal in the dorsal columns with an inverted V pattern on axial T2 imaging.
Mobility and functional deficits (Activity limitations): 79 of the 88 cases exhibited some degree of mobility or functional impairment on presentation. Of this, 67% had impairments in mobility alone, 1% had functional impairments alone and 32% had impairments in both mobility and function. Mobility impairments included difficulty walking, squatting, running, transfers and climbing stairs. Functional impairments included difficulty with writing, using common utensils such as spoons, forks and chopsticks, dressing and tying shoe laces.
Treatment instituted including need for inpatient rehabilitation prior to discharge
Only 76 cases received treatment post presentation. 85% received B12 supplementation alone. 3% received vitamin B12 and oral methionine, 1% received B12 supplementation and methyl prednisolone, 1% received methylprednisolone alone, 1% received plasmapheresis alone and 1% received intravenous immunoglobulin (IVIG), methylprednisolone and vitamin B12.
30% of the 76 cases who received some form of treatment, also received a period of rehabilitation and 70% did not receive a period of inpatient rehabilitation.
Long-term outcomes post nitrous oxide exposure (Impact on Participation in everyday life situations):
Only 57 cases documented the presence or absence of residual neurological deficits on discharge or on subsequent follow-up. 60% had ongoing deficits while 40% had no residual neurological deficits. Of those who did have residual deficits, 41% had mild and 59% had moderate to severe deficits. This is also illustrated in Figure 4. Mild deficits were characterized as those that had no impact on everyday function such as mild paraesthesia, numbness or weakness. Whereas moderate to severe deficits were classified as those that had an impact on everyday function on discharge. Of those cases with residual deficits, only 40% received a period of rehabilitation, with 60% not offered any rehabilitation intervention prior to discharge.
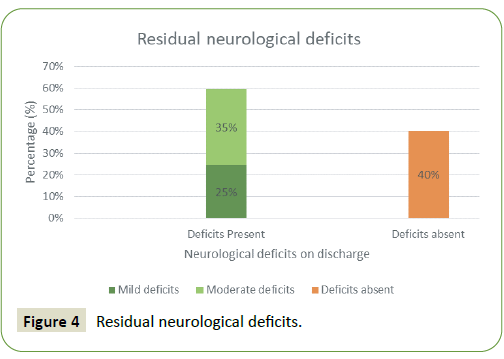
Figure 4: Residual neurological deficits.
Discussion
The neurological deficits that develop following prolonged nitrous oxide exposure is secondary to its effects on the vitamin B12. In the human body vitamin B12 acts as a cofactor in 2 main enzymatic pathways namely methylmalonyl-Coenzyme-A mutase (MMCoAM) and 5-methyltetrahydrofolate-homocysteine methyltransferase (MTR) which are both key in myelin and DNA synthesis. Vitamin B12 deficiency results in disruption of both these pathways leading to disruption of the structural integrity of myelin leading to the changes associated with B12 related neurological sequelae [12]. In this current review, serum vitamin B12 levels were documented in all cases. Of this >50% demonstrated low serum levels. However, it is to be noted that serum vitamin B12 does not reliably represent cellular B12 levels and it is possible for patients to be deficient of functional B12 despite demonstrating normal serum values. This was consistent with the findings of this review, with some patients demonstrating normal serum vitamin B12 levels but having elevated methylmalonic acid and homocysteine levels which are markers of functional B12 deficiency. Interestingly, only 4 cases in this present review had a history of previous B12 deficiency. This contrasts the findings of a systematic review published looking at complications relating to nitrous oxide in an anaesthetic setting which reports that the majority of patients who did suffer complications post nitrous oxide exposure demonstrated prior subclinical B12 deficiency [13].
The number of papers reviewed were too few to determine if a previous history of vitamin B12 deficiency predisposed to worse outcomes. Given that the majority of the case reports did come from countries of low prevalence rates of vitamin B12 deficiency, this highlights the need for future research to determine if there is greater risk of nitrous oxide associated complications in individuals from high prevalence regions of vitamin B12 deficiency.
There was also very poor reporting of duration, frequency or quantity of nitrous oxide consumed in the available case reports we assessed. However, a previously published experimental rat model demonstrated a time-dependent effect of nitrous oxide on MTR and MMCoAM activity levels which suggests that prolonged use of nitrous oxide has a greater propensity for worse outcomes and a greater likelihood of residual deficits compared to shorter exposure [14]. This brings to light the importance of clinicians cautioning patients who present post recreational nitrous oxide exposure the importance of abstinence from future use.
The most common neurological manifestation in vitamin B12 deficiency is subacute combined degeneration of the spinal cord which is characterized by dorsal column pathology with sensory deficits (mainly paraesthesia and impaired proprioception) which can later progress to lateral corticospinal tract involvement with associated ataxic gait and pyramidal weakness [15]. This was consistent with the findings in this present review with 37 cases documenting a diagnosis of subacute combined degeneration of the spinal cord with associated neurological impairments on presentation and physical examination.
79 of the 88 cases included in this review reported a history of mobility and functional deficits on presentation. This is likely secondary to the sensory and motor disturbance associated with B12 deficiency secondary to nitrous oxide exposure. Interestingly, on discharge only 34 cases reported residual impairments. This is in keeping with the literature to suggest that majority of individuals do improve significantly post abstinence and treatment with B12 supplementation. However, of the 34 cases, 59% had moderate to severe deficits. Inpatient rehabilitation was only offered to a minority of patients with only 76 reports documenting if rehabilitation was even offered. Only 30% of this group was referred to inpatient rehabilitation. A large proportion of patients (60%) with residual deficits on discharge did not receive a period of necessary rehabilitation.
This finding draws our attention to the fact that a multidisciplinary rehabilitation program provided either in the acute hospital or in an inpatient rehabilitation setting remains an unexplored treatment option for this patient group. A structured multidisciplinary rehabilitation program will be able to address many of the common impairments associated with nitrous oxide abuse including gait disturbance, sensory disturbance, autonomic dysfunction including bowel, bladder deficits and sexual dysfunction and cognitive impairments. Residual deficits would have long-term implications on community reintegration activities including return to driving, working and studying which can have significant implications on long-term quality of life. This is best addressed by a multidisciplinary assessment and rehabilitation plan.
Zhao, et al. [16] detailed 2 patients (18 and 21 years) presenting with symptoms consistent with subacute combined degeneration of the spinal cord post chronic nitrous oxide abuse with associated deficits in mobility and function including difficulty walking, climbing stairs and squatting. Both cases were referred for a period of inpatient rehabilitation and upon discharge had recovered fully with no residual impairments. The importance of a rehabilitation program to be part of the treatment regimen for this patient group is even more imperative given that these individuals are significantly younger compared to the usual patient group with vitamin B12 deficiency who tend to be significantly older. The authors suggest on admission to an acute hospital setting these patients should be reviewed by the ward allied health team and referred early to the local rehabilitation service. This will allow planning of ongoing therapy whether in an inpatient or outpatient setting.
We acknowledge several limitations in this present systematic review. Firstly, all included articles were case studies and small case series. This carries a high degree of bias and thus the results of this present review have to be interpreted cautiously. However, the nature of the condition does not render itself to more highquality study types such as RCTs. Secondly; we researched the available databases in a retrospective manner which has the possibility of missing suitable articles. Thirdly we did not search the grey literature which can potentially miss unpublished works relating to the topic. Fourth our current review did not have enough statistical power due to poor reporting to render itself for statistical analysis including a multivariate analysis which could potentially demonstrate associations between differing clinical variables and patient outcomes.
Conclusion
Even though the use of Nitrous oxide has been in steady decline in the field of anaesthetics, it has made a worrisome resurgence in the recreational setting particularly popularized as “Whippets” or “Hippy Crack” in clubs and university parties. In many countries including Australia and the UK, it remains legal, cheap and readily available. Given its relative lack of withdrawal or dependence its inherent risks remain grossly underestimated. Based on the findings of this review a multidisciplinary rehabilitation program remains a relatively unexplored treatment option for this patient group.
40336
References
- Randhawa G, Bodenham A (2016) The increasing recreational use ofnnitrous oxide: history revisited. Br J Anaesth 116: 321-324.
- Lassen HC, Henriksen E, Neukirch F, Kristensen HS (1956) Treatmentnof tetanus: Severe bone-marrow depression after prolonged nitrousoxidenanaesthesia. Lancet 270: 527-530.
- Garakani A, Jaffe RJ, Savla D, Welch AK, Protin CA, et al. (2016)nNeurologic, psychiatric, and other medical manifestations of nitrousnoxide abuse: A systematic review of the case literature. Am J Addictn25: 358-269
- Winstock A, Barratt M, Ferris J, Maier L (2017) Global Drug Surveyn2017. London: Global Drug Survey.
- Amsterdam JV, Nabben T, Brink WV (2015) Recreational nitrous oxidenuse: Prevalence and risks. Regul Toxicol Pharmacol 73: 790-796.
- Oussalah A, Julien M, Levy J, Hajjar O, Franczak C, et al. (2019) Globalnburden related to nitrous oxide exposure in medical and recreationalnsettings: A systematic review and individual patient data metaanalysis.nJ Clin Med 8: 1-18.
- The Joanna Briggs Institute (2015) The Joanna Briggs InstitutenReviewers’ Manual 2015: Methodology for JBI Scoping Reviews.nAdelaide: The Joanna Briggs Institute.
- Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reportingnitems for systematic reviews and meta-analyses: The PRISMAnstatement. PLOS Medicine 6: 1-6.
- Joanna Briggs Institute. (2017). Joanna Briggs Institute CriticalnAppraisal Tools. Retrieved from Joanna Briggs Institute: https://njoannabriggs.org/critical-appraisal-tools
- Keddie S, Adams A, Kelso A, Turner B, Schmiere K, et al. (2018) Nonlaughing matter: subacute degeneration of the spinal cord due tonnitrous oxide inhalation. J Neurol 265: 1089-1095.
- Lin R, Chen H, Chang Y, Su J (2011) Subacute combined degenerationncaused of nitrous oxide intoxication: Case reports. Acta NeurologicanTaiwanica 20: 129-137.
- Hathout L, El-Saden S (2011) Nitrous oxide-induced B12 deficiencynmyelopathy: Perspectives on the clinical biochemistry of vitaminnB12. J Neurolo Sci 301: 1-8.
- Patel K, Munne J, Gunness V, Hersey D, Alshafai N, et al. (2018)nSubacute combined degeneration of the spinal cord followingnnitrous oxide anesthesia: A systematic review of cases. Clin NeurolnNeurosurg 173: 163-168.
- Kondo H, Osborne M, Kolhouse J, Binder M, Podell E, et al. (1981)nNitrous oxide has multiple deleterious effects on cobalaminnmetabolism and causes decreases in activities of both mammalianncobalamin-dependent enzymes in rats. J Clin Investig 67: 1270-1283.
- Antonucci M (2018) Subacute combined degeneration fromnrecreational nitrous oxide inhalation. J Emerg Med 54: 105-107.
- Zhao B, Zhao L, Li Z, Zhao R (2020) Subacute combined degenerationninduced by nitrous oxide inhalation. Medicine 99: 1-4.









