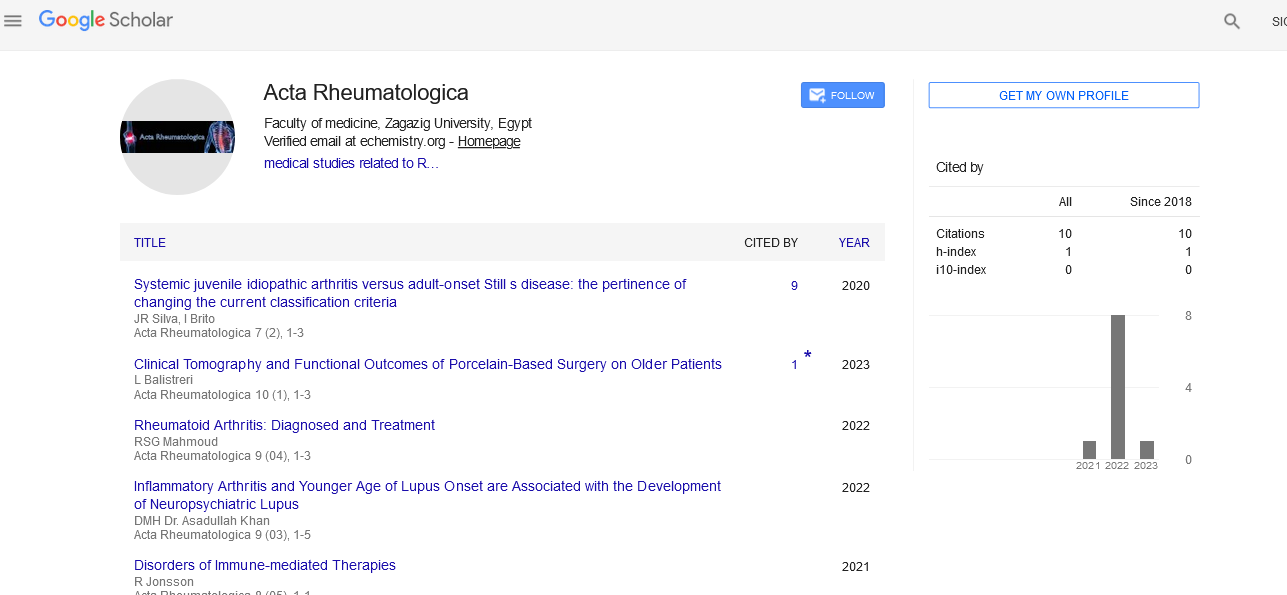
Opinion - (2022) Volume 9, Issue 4
Significance of Monocytes & Macrophages in Spondyloarthritis
Ramel Jonsson*
National University Hospital of Iceland, Reykjavik, Iceland
*Correspondence:
Ramel Jonsson, National University Hospital of Iceland, Reykjavik,
Iceland,
Email:
Received: 03-Aug-2022, Manuscript No. ipar-22-12502;
Editor assigned: 05-Aug-2022, Pre QC No. ipar-22-12502 (PQ);
Reviewed: 23-Aug-2022, QC No. ipar-22-12502;
Revised: 24-Aug-2022, Manuscript No. ipar-22-12502 (Q);
Published:
31-Aug-2022, DOI: 10.36648/ipar.22.9.4.11
Opinion
Spondyloarthritis (SpA) is a collection of chronic inflammatory
illnesses that mostly affect the spine and joints, with the sacroiliac
joint being the most commonly implicated [1]. These diseases,
which affect about 1% of the global population, cause serious
ailments, suffering, disability, resulting in significant health
and economical concerns [2]. In contrast to other rheumatic
diseases such as rheumatoid arthritis (RA) and systemic lupus
erythematosus (SLE), the prevalence of spondylarthritis is
equivalent in males and females, and the disease's beginning
(third-fourth decade) occurs sooner than in other rheumatic
joint diseases. The innate immune system cells are the initial
line of defence against infections. Mucosal-associated invariant T
(MAIT) cells, invariant natural killer T (iNKT) cells, gamma delta T
cells (T cells), innate lymphoid cells (ILCs), neutrophils, mast cells,
eosinophils, and monocytes and macrophages are among these
cell types [3]. Monocytes and macrophages play an important
role in the activation of the innate immune system, releasing
inflammatory cytokines such as TNF, IL-6, IL-1, and chemokines
after pathogen detection, which activate and attract other
immune cells to the inflammation sites [4].
In the pathophysiology of SpA, a genetic relationship has been
demonstrated. The strongest link was discovered in the HLA-B27
gene, which encodes the human leukocyte antigen (HLA) class
I molecule 27 (HLA-B27), a key risk factor for the development
of SpA, particularly AS and USpA [5]. Human leukocyte antigens
(HLA), also known as Major Histocompatibility Complex (MHC),
are in charge of presenting intracellular and extracellular
peptides to immune system cells for activation. HLA-B27 is an
MHC class I molecule that presents intracellular peptides to
CD8+ T lymphocytes [6]. A number of epigenetic imbalances
have also been linked to SpA development. Methylation is an
important epigenetic mechanism involved in several illnesses,
including SpA. Aberrant methylation is caused by an increase in
the addition of methyl groups in specific gene regions. As a result,
hypermethylation of promoter regions reduces gene expression,
frequently exacerbating pathogenic symptoms [7]. In AS patients,
GWAS studies revealed differentially methylation sites in the
HLA-DQB1 gene. MicroRNAs (miRNAs) are also key epigenetic
factors, and several studies have found that various miRNAs are dysregulated in SpA patients. One of the most significant studies
discovered a pattern of 13 miRNAs unregulated in monocytes
and 11 miRNAs deregulated in CD4+ T cells in patients with
axSpA compared to controls, both of which are implicated in
the pathogenesis of SpA. There are now several hypotheses that
can coexist and all agree that the trigger for autoinflammatory
processes is mediated by the HLA-B27 antigen. The first hypothesis
holds that particular HLA-27 subtypes bind to peptides identified
by CD8+ cells, activating autoreactive T cells [8].
The second hypothesis proposes that defective HLA-B27 folding
at the endoplasmic reticulum of immune cells activates the
unfolding protein response (UPR) pathway, which induces the
translocation and thus activation of transcription factor NFkB
to the nucleus, resulting in the production of cytokines
involved in disease pathogenesis by various inflammatory cells.
Innate immune system cells are involved in the beginning and
progression of SpA, and monocytes have been shown to play a
critical role in the pathogenesis of these disorders. Monocytes
are classified into three types based on the expression of the
surface antigens CD14 and CD16. The most prevalent population,
classical monocytes (approximately 90%), express high levels of
CD14 but lack CD16 (CD14++ CD16-).Macrophages are innate
immune system cells that are found in all tissues and body
compartments and act as the initial line of defence against
infection. They are the primary phagocytic cells, but they also
serve as antigen presenters and release cytokines that aid in
immune system activation. Macrophages are also important in
the preservation of tissue homeostasis and in the orchestration
of chronic inflammation seen in a variety of illnesses, including
SpA.
Spondyloarthritis (SpA) refers to a group of chronic inflammatory
illnesses, the most common of which are ankylosing spondylitis
(AS) and psoriatic arthritis (PsA). These disorders share
genetic, clinical, and immunological characteristics, such as the
involvement of human leukocyte antigen (HLA) class I molecule
27 (HLA-B27), peripheral, spine, and sacroiliac joint inflammation,
and the existence of extra-articular symptoms (psoriasis, anterior
uveitis, enthesitis and inflammatory bowel disease).
REFERENCES
- Nichols, Bainton, Dorothy Ford, Farquhar, Marilyn G, et al. (1971) Differentiation of monocytes. Journal of Cell Biology 50: 498-515.
Google Scholar, Crossref
- Zini G (2021) How I investigate difficult cells at the optical microscope. Int J Lab Hematol 43: 346-353.
Google Scholar, Crossref, Indexed at
- Monga I, Kaur K, Dhanda S (2022) Revisiting hematopoiesis: applications of the bulk and single-cell transcriptomics dissecting transcriptional heterogeneity in hematopoietic stem cells. Briefings in Functional Genomics 21: 159-176.
Google Scholar, Crossref, Indexed at
- Ziegler-Heitbrock, HW Loems, Passlick Bernward, Flieger Dimitri (1988) The Monoclonal Antimonocyte Antibody My4 Stains B Lymphocytes and Two Distinct Monocyte Subsets in Human Peripheral Blood. Hybridoma 7: 521-527.
Google Scholar, Crossref, Indexed at
- Passlick Bernward, Flieger Dimitri, Ziegler-Heitbrock, HW Loems (1989) Characterization of a human monocyte subpopulation coexpressing CD14 and CD16 antigens. Blood 74: 2527-2534.
Google Scholar, Crossref, Indexed at
- Ziegler-Heitbrock, Loems (2007) The CD14+ CD16+ blood monocytes: their role in infection and inflammation. J Leukoc Biol 81: 584-592.
Google Scholar, Crossref, Indexed at
- Hofer Thomas P, Van de Loosdrecht Arjan A, Stahl-Hennig Christiane, Cassatella Marco A, Ziegler-Heitbrock Loems, et al. (2019) 6-Sulfo LacNAc (Slan) as a Marker for Non-classical Monocytes. Frontiers in Immunology 10: 2052.
Google Scholar, Crossref, Indexed at
- Ghattas Angie, Griffiths Helen R, Devitt Andrew, Lip Gregory YH, Shantsila Eduard, et al. (2013) Monocytes in Coronary Artery Disease and Atherosclerosis. . Am Coll Cardiol 62: 1541-1551.
Google Scholar, Crossref, Indexed at
Citation: Jonsson R (2022) Significance
of Monocytes & Macrophages in
Spondyloarthritis. Acta Rheuma, Vol. 9 No.4: 11.