Keywords
Regulation disorders of sensory processing; Periventricular hyperyntensities; Neurodevelopmental disorder; Child neurological dysfunctions
Introduction
In developmental neuroscience, the most recent findings confirm the wide plasticity of the nervous system in the early years of life, both in the case of typical development and in the presence of genetic abnormalities or lesions. The plasticity is greater during the so-called "critical periods" in which the environment has a better chance to play a positive influence, but also potentially negative ("maladaptive plasticity") on the development of the nervous system [1]. As regards the scope of clinical research and healthcare organization, these discoveries make it increasingly important the weight of early diagnosis and timely treatment of developmental disabilities, starting from psychomotor and sensory disorders till to those mental and relational.
Research in the field of child mental health has led to an important transformation in the knowledge of early psychopathological disorders.
At the same time, great attention has been paid to the socalled “minor” disorders of perceptual-motor development such as the Developmental Coordination Disorder (DCD) and the disorders of “new definition” such as the Deficit in Attention, Motor control and Perception (DAMP) and the Regulation Disorders of Sensory Processing (RDSP), attributable in most, if not in all cases, to a dysfunction in the processing and/or integration of sensory-perceptive information [2]. This aspect refers to the concepts of "connectivity" and "system", to the extent that it does not require a macroscopic lesion to the onset of the disorders, but it is quite conceivable a malfunction of circuits and bundles of intra- and interhemispheric fibers that underlie them [3].
In 2002, Hadders-Algra’s team published an interesting work on minor neurological dysfunctions asserting the certain existence of a correlation between these and adverse perinatal factors, the description of which, however, remained unspecific [4].
The current hypothesis is that a minimal brain damage origins the minimum brain dysfunction that determines the minor neurological dysfunction. The questions that remain unresolved are: what is meant by minimal brain damage and/or dysfunction? How much the individual vulnerability, presumably genetic, it is crucial in the presentation of a clinical framework rather than another for the same period of onset and topography of the lesion? Moreover, how much the environment affects through epigenetic mechanisms? Currently the majority of neuroradiological and clinical neuropsychiatric studies are aimed at identifying intensiometric and morphological features which allow to associate specific neuroimaging characteristics to certain developmental disorders, with the ultimate goal of increasing the predictability of injuries and the anticipate both diagnosis and therapy [5-7]. At the same time great attention is paid to the research of the so-called "susceptibility genes" that are typical of certain diseases [8]. In the context of neurodevelopmental disorders lies a clinical framework whose diagnosis is steadily increasing, that is the Regulation Disorders [9].
As part of neurodevelopmental disorders, there is another disorder of early childhood, the Regulatory-Sensory Processing Disorder (RSPD) defined on the diagnostic manual DC: 0-3R as "the child's difficulties to regulate his own behaviour, his physiological, sensory, attentional, motor or affective processes and difficulties in organizing a state of calm, of alert or a positive affective state". For the diagnosis must be present sensory processing and motor difficulties and a specific pattern of behaviour, characteristics that allow the definition of the "type" hypersensitive, hyposensitive (with the relative subtypes) or impulsive/disorganized [10].
At the base of the RSPD definition, there are the works of Dr. Anne Jean Ayres, an occupational therapist with a PhD in developmental psychology, who had always worked at the Brain Research Institute at the University of California in Los Angeles. In the course of her research at this institution, she coined the term "sensory integration dysfunction" to describe what she considered the basis of learning disabilities of which she was dealing. These works precisely influenced the sort of the publishing of the diagnostic manual DC: 0-3 and then of its review DC: 03R in which the regulation disorders clearly refer to the abnormal processing and sensory integration. Another sphere of influence concerns the presence and nature of sensory perception disorders in autism spectrum as well as on the development of different methods of treatment of children "with special needs" [11].
Clinical practice has led us to hypothesize an association between "minor" injuries of the white matter (WM) and neurodevelopmental disorders with early onset, which will be the object of the research detailed below.
The main goal of our work was to demonstrate the association between low grade non-cystic diffuse lesions of the periventricular white matter (PVWM) presumably attributable to a prenatal onset and specific neuropsychiatric patterns with onset in early childhood.
The hypothesis is that these abnormalities of the PVWM can determine an alteration in brain connectivity systems with a pathogenic role in certain neurodevelopmental disorders with onset in the first years of life, particularly in RDSP. In favour of this hypothesis, there are many scientific works that, through the use of neuroimaging techniques, such as the highresolution MRI and tractography, show the existence of a dense network of intracerebral fibers and alterations of traits or bundles of fibers corresponding to conventional MRI pictures as diffuse hyperintensity of the PVWM in FLAIR sequences (Fluid Attenuated Inversion Recovery). FLAIR is a pulse sequence used in MRI that allow to visualize the brain in T2 weighted images and to suppress cerebrospinal fluid effects on the images, so it is used to bring out the periventricular hyperintense lesions.
It also seems interesting to speculate that early insults, presumably hypoxic / ischemic, borne by the subplate can determine not so much macroscopical alterations but rather functional, affecting multiple neural circuits that in the subplate neurons find an important relay point between thalamic, brainstem and subcortical afferent fibers and mainly directed to the cortex efferent fibers.
Methods
Participants
We conducted a prospective clinical and neuroradiological study of 20 children (15 males and 5 females) aged between 2 and 4 years (mean = 25.5; SD = 6.5). The sample was divided into children with RDSP (N = 10) and healthy children (N = 10). All children were born between the 37th and 40th gestational week, with a birth weight appropriate for gestational age. None of the children had a documented perinatal distress, with an Apgar index ranging from 8 to 10 at the first and the fifth minute.
Procedures
The diagnosis of RDSP was formulated according to the DC: 0-3R, by excluding specific visual or hearing pathologies in accordance with the inclusion criteria; the clinical evaluation was integrated with neurophysiological tests: the visual and acoustic Evoked Potentials (EP) of the sperimental group of children resulted affected in most cases by a modest increase of latencies, while in only 1 child epileptiform EEG abnormalities were present. In healthy children no neurophysiological anomaly was found.
All the MRI analyses were performed using the RM Philips Full 1.5 T equipment; the images were acquired in SE technical (spin-echo) and FAST-SE with multiplanar axial, sagittal and coronal weightings in T1 and T2, with and without infusion of paramagnetic contrast.
Results
As shown in Table 1, in 8 of the 10 children with RDSP the MRI revealed the presence of a low grade non-cystic diffuse hyperintensity of the periventricular white matter typically noticeable in FLAIR sequences, not associated with morphological and structural abnormalities of the lateral ventricles (LV); this value was significantly higher than that found in the group of healthy children (p<0.001). In 5 of the 8 children the PVHs showed a posterior-anterior grading, with involvement of the deep temporo-parietal area in 2 of them; in 6 of the 8 children with positive MRI, the PVHs was associated with hypoplasia of the corpus callosum. In none of the healthy children these anomalies were found. In Table 1 the information of both groups are shown.
| |
RDSP children |
Healthy children |
| MRI Normal Findings |
N = 2 |
N = 10 |
| MRI Pathologic Findings |
N = 8 |
N = 0 |
Typology of MRI Pathologic Findings
Patient 1 |
PVH (> post.); CCH |
| Patient 2 |
PVH (> ant.); CCH |
| Patient 3 |
PVH (>temporo-parietal) |
| Patient 4 |
PVH (ant.) |
| Patient 5 |
PVH; CCH |
| Patient 6 |
PVH (>temporo-parietal); CCH |
| Patient 7 |
PVH (> post.); CCH |
| Patient 8 |
PVH (> post.); CCH |
| RDSP: Regulation Disorders of the Sensory Processing;PVH: Periventricular Hyperintensity;CCH: Corpus Callosum Hypoplasia. |
Table 1: MRI normal and pathologic findings of the sample, with the specific typology of pathologic findings.
Figures 1 to 4, show some of the findings highlighted in 3 children of the RDSP group and in 1 of the control group children (Figure 4).
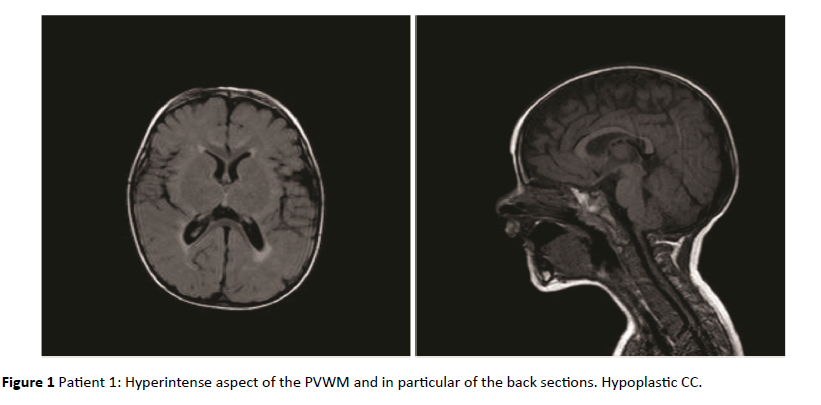
Figure 1: Patient 1: Hyperintense aspect of the PVWM and in particular of the back sections. Hypoplastic CC.
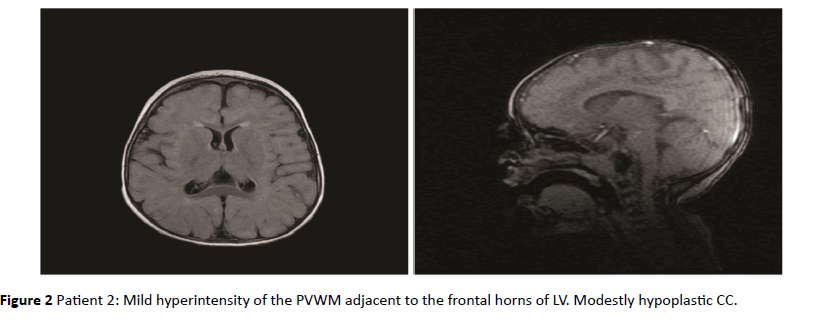
Figure 2: Patient 2: Mild hyperintensity of the PVWM adjacent to the frontal horns of LV. Modestly hypoplastic CC.
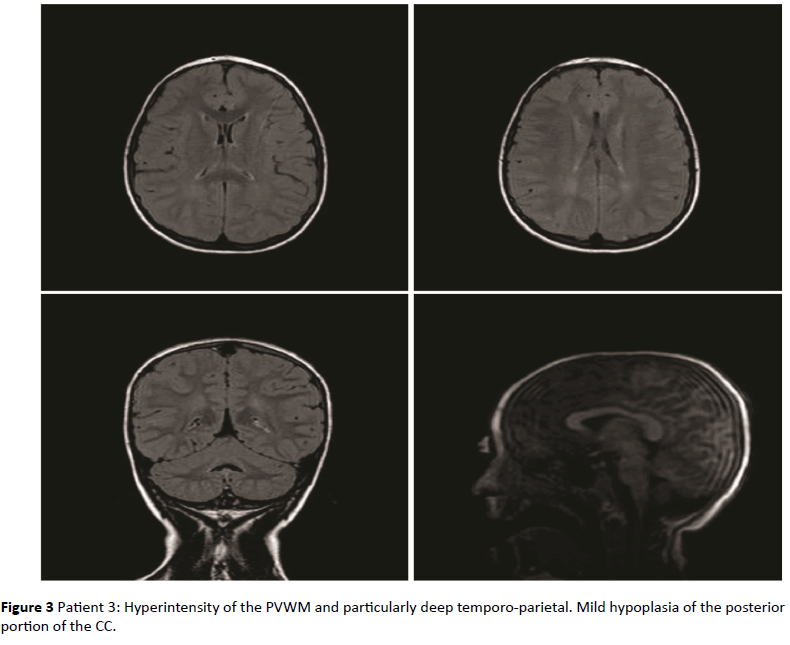
Figure 3: Patient 3: Hyperintensity of the PVWM and particularly deep temporo-parietal. Mild hypoplasia of the posterior portion of the CC.
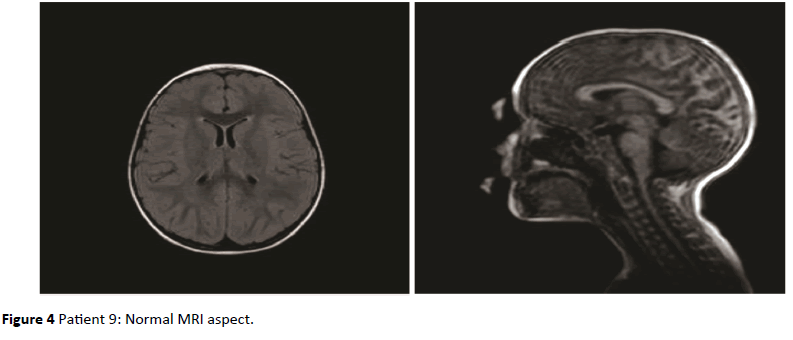
Figure 4: Patient 9: Normal MRI aspect.
Discussion
The RDSP is a neurodevelopmental disorder with onset in early childhood. In literature, there is only one study [12] in which the association between the RDSP and neuroimaging is investigated. Owen and colleagues explored the role of white matter microstructural abnormalities in Sensory Processing Disorders (SPD) using Diffusion Tensor Imaging (DTI), finding a significant decrease in Fractional Anisotropy (FA) and increase in Mean Diffusivity (MD) and Radial Diffusivity (RD) in the SPD cohort compared to controls, primarily involving posterior white matter including the posterior corpus callosum, posterior corona radiata and posterior thalamic radiations. These findings were correlated with statistical significance with behavioural measures, particularly with auditory, multisensory and inattention scores.
These data are in line with what we found through MRI pictures and the hypotheses about anatomical and functional correlations that will be stated below. Compared with Owen’s work, whose sample consists of children aged 8 to 11 years, our study focuses on pre-school children, according to the concepts of early diagnosis and plasticity. To have found a statistical significance between behavioral pattern attributable to RSPD and neuroimaging data is, in our view, an important starting point for further studies on the possible neurobiological or epigenetic vulnerabilities of some children respect to RSPDs but, in terms of development, especially on the positive effects of a therapy centered on the specific needs of each child.
Therefore only few data concerning the neuroanatomical and neurofunctional basis of the disorder are currently available.
Recent studies related to other neurodevelopmental pathologies, such as Autism Spectrum Disorders (ASD) and intellectual disabilities of genetic origin, have shown the presence of alterations in the neural connectivity systems [13,14], as was the case in the epileptologic field with the new definition of “system epilepsy” (SystE) [15].
The findings of our study revealed that the RDSP may be associated with periventricular hyperyntensities (PVHs), and in our sample this happens in the 80% of cases. According to the results of the research in field of neuroscience and neuropathology about the relation between psychiatric diseases and PVHs, it is hypothesized that PVHs could be due to one of the three main causes: ependymal loss, differing degrees of myelination in adjacent fibre tracts and cerebral ischaemia with associated demyelination [16].
A more accurate analysis of the topography of the WM next to LV paves the way for anatomical and functional correlation studies in our opinion very interesting:
• The lateral portion of the frontal horn of LV is covered by inferior fronto-occipital fasciculus (IFOF) that ‘can be considered as a “multi-function” bundle, with each anatomical subcomponent subserving different brain processing The superficial layer and the posterior component of the deep layer, which connects the occipital extra-striate, temporo-basal and inferior frontal cortices, might subserve semantic processing. The middle component of the deep layer could play a role in multimodal sensory–motor integration. Finally, the anterior component of the deep layer might be involved in emotional and behavioural aspects’ [17];
• The upper portion of the frontal horn of LV is covered by the corpus callosum (CC), that is the major neural pathway that connects homologous cortical areas of the two cerebral hemispheres [18];
• The body of the LV is laterally covered by the fibers of the internal capsule formed by bundles of fibers called “projection fibers” because they start from or arrive to the cortex, leading informations from thalamus and basal ganglia, with the function to control the voluntary motor skills and receive the sensitivity of the body, and from the arcuate fasciculus, which connects the areas of Broca and Wernike and transmits auditory-verbal messages after they have reached the associative parietal-temporal-occipital area; frontal-subcortical cognitive and limbic feedback loops modulate higher cognitive functioning [19];
• The lower portion of the LV is in relation to the body of fornix, which is an interhemispheric commissural formation in the shape of a "C" that puts the hippocampus in communion with the mammillary bodies and then with the thalamic nuclei and the cingulate gyrus (posterior fibers) and the amygdala (anterior fibers); it is involved in cognitive functions such as memory and complex visual perception;
• The temporal horn of LV is covered by optical radiation in the depths of the middle temporal gyrus. The auditory radiations cross the optical radiations to the level of the roof of the lower horn.
It therefore seems clear the importance of the bundles of periventricular, associative and commissural fibers in sensoryperceptual processing and integration, that is the basis for multiple functions including learning, visual-spatial, motor planning, social behaviour and language.
The second interesting fact is that in children with RDSP we analysed, we found a posterior-anterior gradient in the localization of the altered PVWM areas, in two cases with involvement of the deep WM of temporal and parietal areas, and this seems to be in accordance with the estimated dysfunction of the associative posterior and limbic areas, with an inevitable impact on the child's emotional sphere too [20].
Moreover, the fact that the widespread periventricular hyperintensity was not associated with morphological and structural abnormalities of the LV gives evidence of a prenatal origin of the damage, in accordance with the hypothesis that intrauterine repeated insults in early gestational ages can determine a damage to the WM (being affected the precursors of oligodendrocytes and potentially the IVZ) of subplate and neural circuits that depend on it [21].
In particular, the intensiometric characteristics (non-cystic widespread hyperintensity of the PVWM in FLAIR images) lay for gliotic lesions with loss of oligodendrocytes and axonal damage.
It should also be remembered that the neurons of the subplate show both glutamatergic and GABAergic activity and are involved in the early formation of functional circuits not only at a local level of the subplate itself, but also at a distance with cortex and thalamus.
Although the subplate is a transitional structure, which will gradually disappear after the birth, its role is crucial as it forms a hub between afferent projections from the brainstem, the thalamus and subcortical nuclei and efferent projections mainly addressed to the thalamus, the cortex and the spinal cord; however, most of the thalamic axons not establish synapses at the level of subplate, remaining "on hold" to enter the cortical plate around the 24th GW [22].
A very interesting fact is that some thalamocortical fibers move to the level of cortical plaque in the visual,somatosensory, auditory and associative areas constituting a connectivity "sensory-driven" framework, while others remain engaged in the formation of endogenous circuits at the level of the subplate. The latter undergoes to a gradual disappearance around the 36th GW; however, it is present as a band below the cortical VI layer and represents an important indicator of the growth of corticocortical and callosal circuits [23].
It seems plausible to assume that insults to the subplate can thus determine an alteration in the formation of multiple neural circuits that remain over its disappearance.
Finally also the finding of hypoplasia of the corpus callosum, and in particular of the posterior third, is in accordance with the clinical presentation of our patients, as in the Witelson division [24], it would contain the connecting fibers between the upper temporal, parietal and occipital regions closely related with auditory-verbal and visual-perceptual functions.
Our hypothesis, which deserves functional neuroimaging and fiber tracking investigations, then is that intensiometric alterations of the WM, and in particular the non-cystic periventricular leukomalacia should be underpinned by abnormalities borne by the intra and interhemispheric connection fibers so determining the formation of disfunctional circuits which above all alter the perception and sensory integration processes. This leads to the inevitable consequences on motor planning and control, but also and especially on the behaviour on the relationship and emotion.
In fact, complex cognitive-emotional behaviours have their basis in dynamic coalitions of networks of brain areas, none of which should be conceptualized as specifically affective or cognitive. ‘Central to cognitive-emotional interactions are brain areas with a high degree of connectivity, called hubs, which are critical for regulating the flow and integration of information between regions [25].
The future perspective is to integrate the instrumental studies with single-voxel spectroscopy evaluation and with the application of the diffusion fiber tracking tensor in order to identify quantitative and quality changes of bundles of fibers and of the specific areas affected by the damage in relation to the clinical presentation and as markers that support the diagnosis [26].
9560
References
- Nava E, Roder B (2011) Adaptation and maladaptation insights from brain plasticity. Prog Brain Res pp: 177.
- Gibbs J, Appleton J, Appleton R (2007) Review: Dyspraxia or developmental coordination disorder? Unravelling the enigma. Arch Dis Child 92: 534-539.
- Gao W, Lin W, Grewen K, Gilmore JH (2016) Functional connectivity of the infant human brain. Plastic and modifiable. Neuroscientist p.1073858416635986.
- HaddersAM (2002) Two distinct forms of minor neurological dysfunction: perspectives emerging from a review of data of the Groningen Perinatal Project. Dev Med Child Neurol 44: 561-571.
- Counsell SJ, Shen Y, Boardman JP, Larkman DJ, Kapellou O, et al. (2006) Changes on magnetic resonance imaging at term-equivalent age axial and radial diffusivity in preterm infants who have diffuse white matter. Pediatrics 117: 376-386.
- Murray ME, Senjem ML, Petersen RC, Hollman JH, Preboske GM, et al. (2010) Functional impact of white matter hyperintensities in cognitively normal elderly subjects. Arch Neurol 67: 1379-1385.
- Cornette LG, Tanner SF, Ramenghi LA, Miall LS, Childs AM, et al. (2002) Magnetic resonance imaging of the infant brain: anatomical characteristics and clinical significance of punctate lesions. Arch Dis Child Fetal Neonatal 86: 171-177.
- Degangi GA, Breinbauer C, Roosevelt JD, Porges S, Greenspan S (2000) Prediction of childhood problems at three years in children experiencing disorders of regulation during infancy. Infant Mental Health Journal 21: 156-175.
- Ellis BJ, Boyce WT, Belsky J, Bakermans-Kranenburg MJ, van Ijzendoorn MH (2011) Differential susceptibility to the environment: An evolutionary–neurodevelopmental theory. Development and Psychopathology.
- Zero to three (2005) Diagnostic classification of mental health and developmental disorders in infancy and early childhood, (revisededn.)Zero to Three Press. Washington. D.C.
- Ayres AJ, Robbins J (2005) Sensory integration and the child: understanding hidden sensory challenges. Western Psychological Services.
- Owen JP, Marco EJ, Desai S, Fourie E, Harris J, et al. (2013)Abnormal white matter microstructure in children with sensory processing disorders. NeuroimageClin 2: 844-853.
- Tebartz van Elst L, Riedel A, Maier S (2016) Autism as a disorder of altered global functional and structural connectivity. Biol Psychiatry 79: 626-627.
- Dean Iii DC, Travers B, Adluru N, Tromp DP, Destiche D, et al. (2016) Investigating the microstructural correlation of white matter in autism spectrum disorder. Brain Connect.
- Avanzini G, Manganotti P, Meletti S, Moshe SL, Panzica F, et al. (2012) The system epilepsies: a pathophysiological hypothesis. Epilepsia 53: 771-778.
- Thomasa AJ, O’Briena JT, Barbera R, McMeekinb W, Perryc R (2003) A neuropathological study of periventricular white matter hyperintensities in major depression. Journal of Affective Disorders 76: 49-54.
- Sarubbo S, De Benedictis A, Maldonado IL, Basso G, Duffau H (2013) Frontal terminations for the inferior fronto-occipital fascicle: anatomical dissection, DTI study and functional considerations on a multi-component bundle. Brain Structure and Function 218: 21-37.
- Bloom JS, Hynd GW (2005) The role of the corpus callosum in interhemispheric transfer of information: excitation or inhibition? Neuropsychol Rev 15: 59-71.
- Levitt JJ, Kubicki M, Nestor PG, ErsnerHH, Westin CF, et al. (2010) A diffusion tensor imaging study of the anterior limb of the internal capsule in schizophrenia. Psychiatry Res 184: 143-150.
- Viviani R (2013) Emotion regulation, attention to emotion, and the ventral attentional network. Front Hum Neurosci.
- Pogledic I, Kostovic I, FalletBC, AdleBH, Gressens P, et al. (2014) Involvement of the subplate zone in preterm infants with periventricular white matter injury. Brain Pathol 24: 128-141.
- Kostovic I, Rakic P (1990) Developmental history of the transient subplate zone in the visual and somatosensory cortex of the macaque monkey and human brain. J CompNeurol 297: 441-470.
- KostoviI, Judas M (2007) Transient patterns of cortical lamination during prenatal life: do they have implications for treatment? NeurosciBiobehav Rev 31: 1157-1168.
- Witelson SF (1989) Hand and sex differences in the isthmus and genu of human corpus callosum. A postmortem morphological study. Brain 112: 799-835.
- Pessoa L (2008) On the relationship between emotion and cognition. Nat Rev Neurosci 9: 148-158.
- American Psychiatric Association (2013). Diagnostic and statistical manual of mental disorders (5thedn.). Washington, DC.









