Keywords
|
| Neurological disorders; Alzheimer; Marine carbohydrates; Heparan sulfate; Sulfated chitosan |
Introduction
|
| Neurodegenerative disorders (ND) are a heterogeneous group of disorders, increasingly prevalent among the elderly people and presents a very key challenge to researchers and health care industries. ND includes AD, Parkinson’s (PD), Huntington’s (HD), Amyotrophic lateral sclerosis (ALS) and prion diseases are the major ND which shares common pathological phenotype, causing neurological dysfunction and loss of neuronal cells in brain [1,2]. They are characterized by the aggregation of misfolded proteins, which trigger a cascade of molecular events leading to accumulation of certain peptides and proteins such as amyloid β (Aβ), huntingtin, α-synuclein etc and concomitant failure of normal biological function. The aggregation of misfolded proteins causes the formation of lewy bodies, tangles and plaques in the brain [3,4]. |
|
AD
|
| AD is one of the progressive, irreversible ND of the central nervous system, most common among the aged population leading to memory loss and progressive cognitive decline. |
| It is characterized by the formation of senile plaques and neurofibrillary tangles, decrease in cholinergic transmission, and psycho-behavior disturbances [5,6]. Although, the cause of disease is still unclear, multiple factors are thought to play a deleterious role in the pathogenesis including abnormal proteins, oxidative stress, excessive metal ion accumulation in brain, depletion of endogenous antioxidants, glutametric neurotoxicity, reduced expression of tropic factors and inflammation causing decrease of neurons and dysfunction of ubiquitin-proteasome systems [7,8]. |
|
Prevalence and current therapy
|
| World health organization (WHO) statistics reports that the prevalence of AD is high with more than 30 million people affected with AD worldwide, and is predicted to increase by 70% to more than thrice in 2050 [9]. The Food and Drug Administration (FDA) has approved two types of medications towards mild and moderate stages of AD (viz. Tacrine, Donepezil, Rivastigmine, Memantine). Majorly, these drugs function as acetylcholinesterase (AChE) inhibitors or target the N-methyl D-aspartate (NMDA) receptors [10]. No effective treatment that modifies or stops the progression of AD is currently available [11]. Most of the currently available drugs from the market are either synthetic or terrestrial based natural products [12]. More than 200 AD drug candidates have failed in later stage of clinical phase studies due to associated adverse effects such as loss of appetite, diarrhea, muscle cramps, nausea, restlessness etc. [7,12]. Hence, the wide range of pathological features in AD and the inability to meliorate the disease condition warrants a growing set of promising therapeutic targets towards AD therapy and management [13,14]. The current review focuses on the importance of carbohydrates from marine derived compounds against ND. |
|
The marine diversity
|
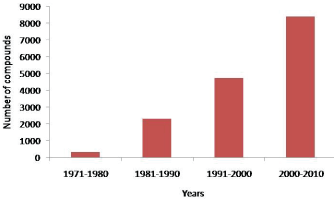
| The ocean covering nearly 70% of the earth area provides a fascinating range of biodiversity of ~2,210,000 species out of which only 190000 are identified so far [15,16]. Marine environment exhibits extremely harsh physical and chemical environmental conditions that drive the researchers’ towards the production of different molecules with unique structural features when compared to the terrestrial ecosystem. Invariably, the land holds up a total of only 12 phyla, whereas the marine stream comprises a total of 32 phyla [17,18]. A recent census shows that the field of marine research transcends across 80 nations, involving more than 2700 scientists, concentrating on diversity, distribution and discovering potential drugs from various marine sources [19]. Moreover, the marine system produces an inexhaustible and rich source of potential natural products with a wide array of nutraceutical, cosmeceutical and unique pharmaceutical activities. More than 30000 compounds have been identified from the marine environment with unique structures and associated pharmaceutical activity [18]. Figure 1 shows the decade-wise increase in the exploration of new bioactive compounds from marine sources from 1971 to 2010. Extensive bioprospecting of the marine fauna, flora and microbes have a huge impact on the biomedical industry, producing small molecules with anti-infective, anti-cancer, anti-inflammatory, analgestic, immuno-modulatory, anti-viral, neuroprotective, antifouling and a range of other biological activities. It proves that the marine reservoir holds infinite pharmacologically promising and exciting drug candidates for human health [20-22]. |
|
Marine glycobiology
|
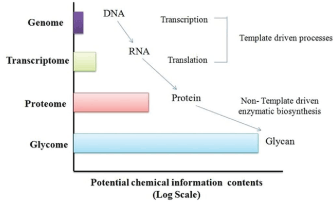
| Marine carbohydrates are the most complex biomolecules in terms of structure. Diversity of monomers, glycosidic linkage, enantiomerism, high dynamic behaviour, anomericity contribute to structural complexity in carbohydrate moieties. Due to the complexity in the carbohydrate structure, they exhibit higher number of classes like N-linked or O-linked oligosaccharides in glycosaminoglycans (GAGs), proteoglycans, sulfated galactans, sulfated fucans etc. [23]. Marine glycobiology offers vast unexploited potential in the discovery of new therapeutics derived from carbohydrates or other molecules which target the biosynthesis and function of saccharides. Glycomics studies are extensively studied in cellular glycoprofiling of cancer and other diseases, glycosylation of therapeutic proteins, correlation between activity and sulfation pattern in GAGs and glycosylation pattern in cell recognition [24]. The structural complexity and multifold diversity of the carbohydrates, in comparison to the protein and nucleic acids is given in Figure 2. |
| Advanced research has boosted the exploration of novel carbohydrate-based drugs by various research groups and multinational pharmaceutical companies for their clinical applications. Glycans from marine origin with peculiar sulfate positions and well defined unique structures have improved their therapeutic properties when compared to terrestrial sources. They possess distinct mechanism which helps to achieve accurate structure-activity relationships. These properties are extremely important for drug discovery development as it constitutes a limited proportion of a larger world of marine therapeutics [23,25-28]. |
| GAGs are long un-branched hetero-polysaccharides, widespread in nature, occurring in a great variety of organisms exhibiting various physiological and biological functions [29-31]. They are majorly classified as sulfated [Keratan sulphate-KS, dermatan sulphate-DS, chondroitin sulphate-CS, heparin-HP and heparan sulphate-HS] and non-sulfated GAGs-[Hyaluronic acid]. Among the GAGs, HP and HS are well explored for their biomedical potential. Commercial heparin obtained for clinical use is isolated from porcine/bovine lung and intestinal tissues. An estimated total of 30 to 40 tons of HP is obtained from 400 to 700 million pigs worldwide [32,33]. Previous reports are available for the management of AD using synthetically and enzymatically obtained HP/HS. Scholefield and coworkers in 2003 [34] have reported the ameliorating effect of HS by inhibiting the amyloid ß secretase (BACE-1). Similarly, HP derived from porcine also has been reported for inhibiting BACE-1 [35]. Leveugle and coworkers in 1994 [36] had demonstrated the interaction of sulfated polymers with Aß, displacing the HS proteoglycan bound to Aß, thereby preventing or slowing amyloid formation and concomitant aggregation. |
|
Limitation of mammalian GAGs
|
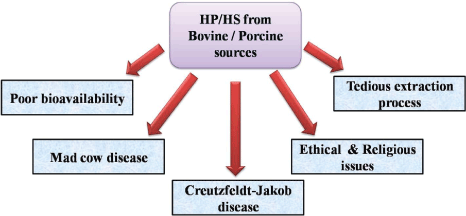
| Uncertainty in distinguishing the source of the HP (bovine or porcine) and difficulties in the maintenance of purity has limited the use of bovine HP (Figure 3). The outbreak of mad cow disease, absence of alternate (chemical, enzymatic & recombinant) HP synthesis practices and ethical constraints warrant the isolation of HP from non-mammalian sources [33,37]. Due to its structural complexity, purification and further characterization of the GAGs is highly challenging [38]. |
|
Marine carbohydrate based drugs for ND
|
| Marine GAGs are capable of binding with a range of proteins with high specificity and actively participate in cell signaling, cell development, cell adhesion, cell differentiation and cell matrix interactions. These biomolecules present a higher potential for medical and pharmaceutical applications against various disorders [39]. Reduced rates of viral infections in marine GAGs favor the utilization of HP and HS from marine sources [33]. Though few reports are available from marine seaweeds (polysaccharides) against ND, therapeutic potential of GAGs from marine animals remain highly limited. The utilization of marine carbohydrate based mimetic compounds, acting as receptor molecules, binding to the misfolded protein aggregates and preventing neuronal damage, is gaining higher importance [40]. The non sulfated GAG, hyaluronan has been associated with an increased occurrence of 26% at the temporal lobe of AD patients, due to binding of the astrocytes to the hyaluronan via their CD44 receptors [41]. HS 971, an oligomannuronate from alginate and heparinoid 971, a derivative of HP obtained from seaweeds screened for their therapeutic ability against AD is currently in phase 11b clinical trials [42]. |
| A series of plant derived polysaccharides, α-glucan-(PC-2, PB-2 and isolichenan) from Parmelia caperata, Flavoparmelia baltimorensis and β-1, 3/1,6 glucan (lentinan) from Lentinula edodes have been reported to exert LTP (Long term potentiation) which is a measure of cellular basis for learning and memory [43-45]. α and ß-glucans enhance hippocampal synaptic plasticity [46]. The sulfated polysaccharide from the brown algae Laminaria japonica, sulfated polymannuroguluronate has been reported to exhibit neuroprotective effect by decreasing apoptosis in PC12 neuronal cells, in vitro [47]. Fucoidan from marine algae such as Fucus vesiculosis, Macrocystis pyrifera and Laminaria japonica has been reported to suppress cellular infiltration of joints [48], neutrophil migration [49] and post-operative pain with no toxicity [50]. Α 1, 4 D glucan from the blue mussel Mytilus coruscus has been reported to exhibit antioxidant activity by preventing liver injury of mice by lowering the levels of serum ALT, AST and hepatic MDA [51]. Cheng and colleagues in 2010 [52] have reported the scavenging abilities of glucanic polysaccharides from Mytilus edulis. Overcoming the aforementioned difficulties, we have isolated the sulfated polysaccharides, HS from the marine scallop Amussium pleuronectus and sulfated chitosan [SC] from the cuttlefish Sepia pharaonis. |
| The marine scallop (A. pleuronectus) bivalves are collected from the Mudasalodai landing center, Parangipettai (lat. 11°29′; long. 79°46′ E), Tamil Nadu, India. The samples are defatted and processed to obtain crude glycosaminoglycan (GAG) [53]. The crude GAG is subjected to enzymatic depolymerization to obtain low molecular weight heparan sulfate (LMW-HS) and its purity was determined by PAGE and metachromatic activity. The elemental and sulfur content was estimated by CHNS analysis. The structure of the heparan sulfate was determined by 1H-NMR spectroscopy. The anticoagulant activities of the LMW-HS are determined [54]. |
| Cuttlefishes (S. pharaonis) are collected from the Kasimedu landing center, Tamil Nadu, India (lat. 13°12′; long. 80°29′). The cuttlebones are removed, washed, shadow dried and crushed to a fine powder. The powder is de-proteinized, decolorized and decalcified to obtain chitin. The chitin is deacetylated to obtain chitosan which is sulfated to yield SC. |
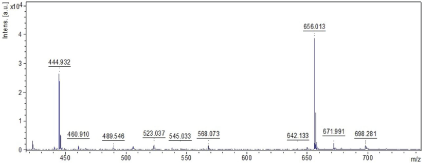
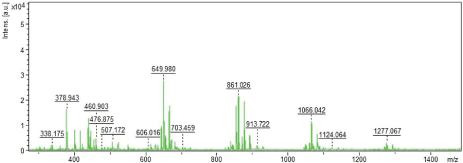
| The molecular weight of the isolated HS and SC are recordeded using Matrix Assisted Laser Desorption-Ionisation-Time of Flight Mass Spectrometry [MALDI-TOF MS]. The molecular weight of HS and SC are determined to be 698 and 1277 Da yielding 21 and 10 ionized fragments respectively (Figures 4 and 5). We have reported the potentials of HS from the marine scallop, A. pleuronectus as an alternate source of anticoagulant [35]. The low molecular weight nature of the sulfated polysaccharides could be hypothesized to render permeability across the bloodbrain barrier. We also hypothesize that the isolated sulfated polysaccharides may competitively inhibit binding of Aß with intrinsic GAG, preventing amyloid formation and aggregation. The sulfated polysaccharides may be further screened for their abilities to function as glyco-mimetics that may prevent the aggregation and neurofibrillar formation, thereby opening novel avenues towards therapy and management of AD. Thus, the sulfated polysaccharides, HS and SC from the marine scallop and cuttlefish could be potential candidates from marine sources towards AD therapy and management in future. |
Conclusion
|
| The mini review discusses the alternative approaches for the therapy and management of ND, essentially affecting the extracellular matrix. The review highlights the promising potentials of carbohydrate based compounds from marine sources with enhanced neuro-biological activity by virtue of its structural features. The structural modifications due to branching and chemical substitutions like sulfation, de-acetylation, size, molecular weight improve binding efficiencies with target proteins associated in ND. These modifications also extend the half-life of the compound increasing its bio-availability. From our study, we expect the LMW-HS from marine scallop and SC from cuttlefish to cross the blood-brain barrier and modulate the action of neurological protein. In purview of the above, carbohydrates from marine sources thus hold immense potentials as an alternate source of anti-alzheimer agents of the future. |
Figures at a glance
|
 |
 |
 |
 |
 |
| Figure 1 |
Figure 2 |
Figure 3 |
Figure 4 |
Figure 5 |
|










