Research Article - (2025) Volume 16, Issue 4
GABA levels decrease in brain areas associated with neurodegeneration in Alzheimer's and Parkinson's diseases: Systematic review and meta analysis
Flavio M. Morais1,
Pollyanna V.G. Silva2 and
Angela M. Ribeiro3*
1Department of Neurosciences, Institute of Biological Sciences, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil, Brasil
2Ministerio Público do Estado de Minas Gerais, Belo Horizonte, Brazil, Brazil
3Department of Biochemistry and Immunology, Institute of Biological Sciences, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil, Brasil
*Correspondence:
Angela M. Ribeiro, Department of Biochemistry and Immunology, Institute of Biological Sciences, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil,
Brasil,
Email:
Received: 10-Jul-2024, Manuscript No. IPJNN-24-15189;
Editor assigned: 12-Jul-2024, Pre QC No. IPJNN-24-15189 (PQ));
Reviewed: 26-Jul-2024, QC No. IPJNN-24-15189;
Revised: 14-Oct-2025, Manuscript No. IPJNN-24-15189 (R);
Published:
21-Oct-2025
Abstract
Background: Data obtained by our group and by other authors directed our interest towards better understanding the involvement of the GABArgic and glutamatergic systems in the neurodegenerative process. We began a more in-depth search into this subject and came across various apparently contradictory data, most likely from the use of different experimental models and measurement conditions. We therefore considered it relevant to carry out a systematic review followed by a meta-analysis of the results, with the aim of answering contradictory questions about the involvement and importance of components of these two systems in the neurodegeneration process associated with AD and PD. There is no systematic survey in the literature aimed at verifying the relevance and degree of significance of the relationships between GABAergic and glutamatergic parameters and the neurodegeneration phenomenon observed in these diseases.
Method: We gathered and analyzed the data obtained from various studies published in the PubMed and Scopus databases between 1987 and 2024, in which the authors measured molecular parameters in samples obtained post-mortem from patients and in experimental models (rodents). The study was divided into two stages. The first stage focused on neurodegenerative processes in a more general way. In this phase, studies were selected in which the measurements were carried out on total brain samples and therefore without specifying the region. The partial analysis of the data obtained in the first stage showed a predominance of studies addressing AD and PD and, in addition, indicated significant effects on decreasing GABA levels. To further explore this result, we decided to add to the keywords of the first stage, the specification of brain regions generally affected in these two conditions and found in studies selected in the first stage. This included the regions of the neocortex, hippocampus, striatum and PAG.
Results: The main findings of the meta-analysis, testing for the overall effect, were that GABA levels in AD are significantly lower in the neocortex of rodents (Z=2.50; p-value=0.01) and humans (Z=3.49; p-value=0.0005) and in the hippocampus of rodents (Z=3.91; p-value<0.0001). This data, decreased GABA was also observed in human whole brain samples (Z=3.66; p-value=0.0003). GABA-A receptor expression was higher in the neocortex (Z=2.47; p-value=0.01) and hippocampus (Z=2.49; p-value=0.01) of affected individuals (rodents). On the other hand, glutamate levels were not affected in either of these two regions, neocortex and hippocampus, in rodents. In Parkinson's disease, GABA levels were lower in the striatum (Z=2.53; p-value=0.01) and PAG (Z=2.59; p-value =0.009) of affected individuals. Unlike AD, there was no difference in GABA levels in the neocortex in humans. There were no differences in glutamate levels in the striatum and neocortex.
Conclusion: based on the data presented here, we found that the literature provides an inconclusive overview of changes in GABA levels and GABAergic components in the brain areas of patients with AD and PD, and therefore grouping the data using the meta-analysis method represents an important contribution of the present study. The data corroborate recent evidence that the original descriptions of neurodegenerative diseases as being mainly due to lesions in dopaminergic systems in the midbrain for PD and hippocampal cholinergic and glutamatergic lesions for AD and other dementias should be updated within a concept of multisystem neurodegeneration, where a progressive functional decline of the GABAergic system needs to be further considered and better studied.
Keywords
GABA; Neurodegeneration; Symptoms; Parkinson's disease; Neocortex
Introduction
The neurobiological bases of the learning and memory process have been intensely investigated by researchers around the world, mainly about spatial learning and memory, which are the cognitive components mainly affected in cases of neurodegenerative diseases. However, despite the efforts of several research groups, much remains unknown about the molecular mechanisms related to these cognitive processes and the processes that result in neuronal loss and cognitive impairment, emotional and motor functions [1-4]. This makes the studies extremely relevant, not only for understanding the cellular and molecular physiology, which serves as the basis for these functions, but also for contributing to the clarification of their dysfunctions, as they occur in neurodegenerative diseases.
Results obtained at our laboratory, using an experimental rodent model, showed that during the spatial learning process there is a significant increase in the levels of the Neurotransmitter γ-Aminobutyric Acid (GABA) in the hippocampus of animals, suggesting a role for this neurotransmitter in the neurobiological mechanism of the learning process [5]. In previous studies, we observed significant correlations between the concentrations of GABA and glutamate and between the concentrations of these neurotransmitters and the performance of animals in the initial phases of the learning process [6]. Our group also showed evidence that impairment in cognitive performance in spatial navigation tasks, induced by insults that cause neurodegeneration (e.g., thiamine deficiency, aging) occurs in the early phase of the learning process and repetition of the task can reverse the deficits initially observed [7-9]. We also observed that thiamine restriction during development induces dysfunctions in spatial cognition and alters GABA and glutamate levels in some brain regions [10]. Using proteomic analysis techniques, we showed that thiamine deficiency increases the expression of the enzyme glutamate decarboxylase, possibly as a compensatory effect, to increase GABA levels reduced by thiamine deficiency, in line with the idea that GABA levels in the hippocampus are relevant to rodent performance in spatial tasks.
Other authors have presented evidence that the imbalance of the GABAergic and glutamatergic systems results in damage to the neurogenesis process and thus, damage to cognitive functions, such as memory [11,12]. Furthermore, data from different studies show that the GABAergic circuit is important for learning and memory [13,14].
Thus, the data obtained by our group and other authors directed our interest to better understand the involvement of excitatory and inhibitory systems in the neurodegenerative process. We began a more in-depth search for this subject and came across several apparently contradictory data in the literature [15,16].
The contradictions most likely come from the use of different experimental models and measurement conditions. Therefore, we consider the relevance of a systematic review study and a meta-analysis of the results, with the purpose of organizing the information and facilitating the understanding and interpretation of the variables, enabling more adequate planning of future studies, through the elaboration of new substantiated questions in the different findings in this field of knowledge, in the last 37 years. Thus, using well-defined criteria and also considering information about the dependent and independent variables, recording the types of models, measurement methods and samples used in each study, we aim to contribute to clarifying the following question: What is the involvement of GABAergic and glutamatergic parameters (e.g. levels of GABA and Glutamate neurotransmitters) in the death of neurons that occurs in neurodegenerative diseases, such as AD and PD?
Materials and Methods
Sample size
Data were collected through a systematic review using the PubMed and Scopus database, from 1987 to 2014, with a search strategy specified by keywords. All data from studies that met the inclusion criteria, presented below, were analyzed. A search in the gray literature was not used.
Inclusion criteria
Studies published until 2024 were included in the analysis, which analyzed components of the GABAergic and/or glutamatergic systems in neurodegenerative processes covering the following aspects:
• Clinical studies
• Experimental animal model
• Primary and organotypic cell cultures
• Original data
Exclusion criteria
Studies in which the data represented duplicate publications by the same author.
Article/study search strategy
The search was carried out using key words in English and articles that met the inclusion and exclusion criteria were selected. The search strategy was divided into 2 stages. In the first stage, a search was made about neurodegenerative processes, more generic, and was based on the following keyword compositions:
• Brain and neurodegeneration and GABA
• Brain and neurodegeneration and glutamate
After this search, We noted the predominance of articles addressing AD and PD and included the keywords in the following compositions, thus constituting the second stage of the present study
• Alzheimer and neurodegeneration and GABA
• Alzheimer and neurodegeneration and glutamate
• Parkinson and neurodegeneration and GABA
• Parkinson and neurodegeneration and glutamate
• GABA and Parkinson and hippocampus
• GABA and Parkinson and striatum
• GABA and Parkinson and cortex
• GABA and Parkinson and PAG
• GABA and Alzheimer and hippocampus
• GABA and Alzheimer and striatum
• GABA and Alzheimer and cortex
• GABA and Alzheimer and PAG
Review methods
Selection of studies (articles): The articles were selected according to the inclusion and exclusion criteria, after an electronic search on PubMed and Scopus, from 1987 to 2024, using the keywords presented above. The selection of articles was made by two reviewers, using a blind method. Any questions related to the inclusion or not of any article/data were discussed with the coordinator, who, using the blind method, decided on the inclusion or exclusion of the data. The same procedure was adopted for data tabulation. The endnote basic virtual version program, from Thomson Reuters, was used to allocate the abstracts and articles selected for the research.
Data extraction
Data from the articles were tabulated in an except spreadsheet, recording the following variables: Article reference, type of study, individual, type of sample (e.g., culture, tissue, post mortem samples from brain patients, brain samples from rodents.), disease/ neurodegenerative process, system components GABA, glutamate, GABA/glutamate (release, uptake, receptors, transporters).
The data extracted from each experimental group were sample sizes, means and standard deviation of the mean. If the study reported the standard error of the mean, this was used to calculate the standard deviation. In some studies, these values were obtained directly from the text. If not explicit, numerical values were calculated from graphs. The sizes of the bars representing the means and standard deviations (or standard errors) of the control and affected groups were measured using a ruler, in mm. Based on the value of the control mean bar and the measurement of this bar in mm using the ruler and the measurement of the experimental group bar in mm using the ruler, a rule of three was used to find the mean value of the experimental group.
Statistical analysis
The selected studies were evaluated through meta-analysis, which constitutes a set of quantitative methods used to contrast and combine data from a systematic review. The weights of the studies are the inverse of the variances of each study. The Rev Man program, version 5.4 was used to carry out the meta-analysis. The studies were grouped seeking homogeneity, separating studies with an experimental rodent model from those with human beings. Studies that referred to the same disease, the same component of the GABA and glutamate system and the same cerebral area were grouped for subsequent statistical analysis.
The estimate of the overall difference between the control and affected (experimental) groups is a weighted average of the individual studies in which the weights are the inverse of the variability of each study. This variability represents the sum of the variance within each study and the measure of heterogeneity between studies.
Heterogeneity was measured and assessed for checking the existence of heterogeneity using Cochran's Q test, the Tau2 measure or Higgins and Thompson's I2 statistic. In the present work, the random effects model and all the tests mentioned above were used to assess the extent of heterogeneity.
The results are presented in Forest Graphs, highlighting the data from the overall effect test, Z values and P values, considering P<0.05 as significant.
Results
In the first stage of the research, two compositions of keywords were used (brain and neurodegeneration and GABA and brain and neurodegeneration and glutamate). As shown in Figure 1 this first phase of searches, 370 articles were analyzed, of which 11 were studies addressing neurodegenerative processes. Of these, 1 article was found about a study of multiple sclerosis in humans, measuring glutamate. There were no other studies of the same type in the first stage, making it impossible to perform a meta-analysis with only one study. Therefore, the meta-analysis of stage 1 was performed with 10 articles. In a second stage, new compositions of keywords were included (Alzheimer's and neurodegeneration and GABA, Alzheimer's and neurodegeneration and glutamate, Parkinson and neurodegeneration and GABA, Parkinson and neurodegeneration and glutamate, GABA and Parkinson and hippocampus; GABA and Parkinson and striatum; GABA and Parkinson and cortex; GABA and Parkinson and PAG; GABA and Alzheimer and hippocampus; GABA and Alzheimer and striatum; GABA and Alzheimer and cortex, and GABA and Alzheimer and PAG). And with these keywords, 517 articles were analyzed. Using the inclusion/exclusion criteria, 42 articles were selected to be included in the meta-analysis, 20 studies addressing AD and 22 studies addressing PD. A significant number of articles were not included, for one or more of the reasons mentioned below:
• Because they are review articles.
• Studies that mention the terms referring to one or more of the compositions of the keywords, but that do not present original data on measures of the variables/parameters mentioned.
• Out-of-scope articles that did not evaluate neurodegenerative processes and components of the GABAergic and/or glutamatergic system.
First, we will present the data obtained in the first stage and then the results of the second stage.

Fig. 1. Flow diagram showing the data relating to the selection of articles in phase 1 and phase 2 of the systematic review
In some meta-analyses, data from the same article are recorded more than once (column 1 of each figure), because the authors performed more than one type of study/comparison between the control and experimental groups.
The studies were grouped in search of homogeneity, with the separation of studies with rodent animal models from those with human beings. Studies that referred to neurodegenerative processes (1st stage) and the same disease (Alzheimer's or Parkinson's, 2nd stage) and to the same component of the GABA and glutamate system (1st and 2nd stages) and to the same brain areas (2nd stage) were grouped (Figure 2).

Fig. 2. Data from the meta-analysis of studies grouped using the keywords ‘Brain and GABA and neurodegeneration’. GABA levels in whole brain samples of rodents. Test for overall effect, Z=0.85 (P=0.40).
The meta-analysis was performed with data obtained from 4 articles (6 studies), comparing GABA levels in rodent experimental models between the control and experimental groups. There is no significant difference between the groups, since the p-value is greater than 0.05 (Z=0.85; p-value=0.40), and the confidence interval includes zero (Figure 3).

Fig. 3. Data from the meta-analysis of studies grouped using the keywords ‘Brain and GABA and neurodegeneration’. GABA-A receptor expression in whole brain samples of rodents. Test for overall effect, Z=2.71 (P=0.0.07).
The meta-analysis was performed with data obtained from 2 articles (6 studies), comparing GABA-A receptor expression in experimental models of Alzheimer's disease between the control and experimental groups, considering the rodent animal model. There is a significant difference between the groups, since the p-value is less than 0.05 (Z=2.71; p-value=0.007), indicating that GABA-A expression is higher in affected individuals (Figure 4).

Fig. 4. Data from the meta-analysis of studies grouped using the keywords ‘Brain and GABA and neurodegeneration’. GABA levels in whole brain post-mortem samples. Test for overall effect, Z=3.66 (P=0.0.0003).
The meta-analysis was performed with data obtained from 2 articles (8 studies), comparing GABA levels in postmortem samples obtained from patients diagnosed with Alzheimer's disease, between the control and experimental (human) groups. There is a significant difference between the groups, since the p-value is less than 0.05 (Z=3.66; p-value=0.0003), indicating that GABA levels are lower in the cortex in affected individuals (Figure 5).
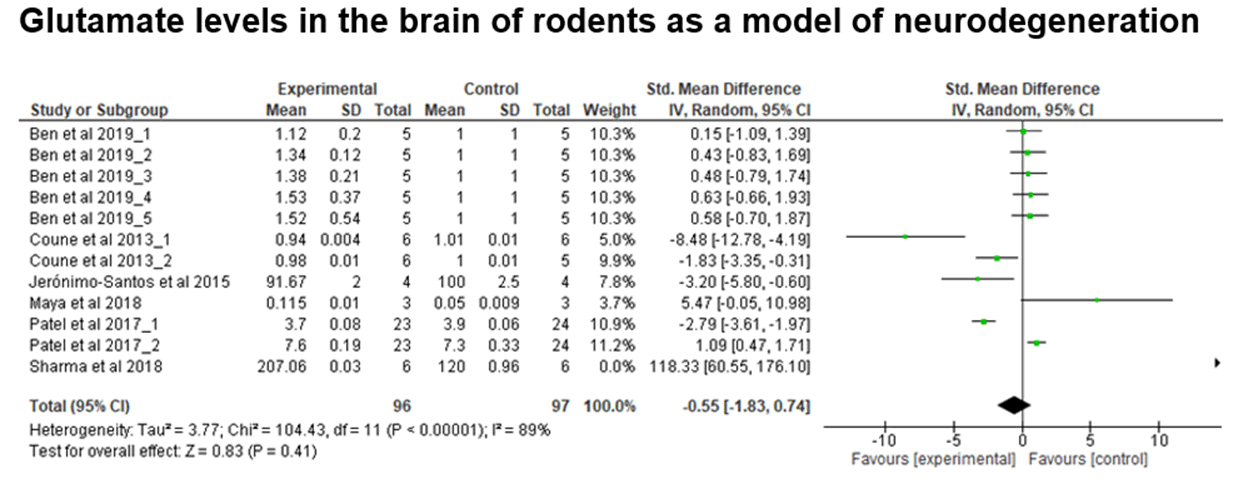
Fig. 5. Data from the meta-analysis of studies grouped using the keywords ‘Brain and Glutamate and neurodegeneration’. GABA levels in whole brain samples of rodents. Test for overall effect, Z=0.83 (P=0.41).
The meta-analysis was performed with data obtained from 6 articles (12 studies), comparing glutamate levels in experimental models between the control and experimental groups, rodent model. There is no significant difference between the groups, since the p-value is greater than 0.05 (Z=0.83; p-value=0.41), and the confidence interval includes zero (Figure 6).
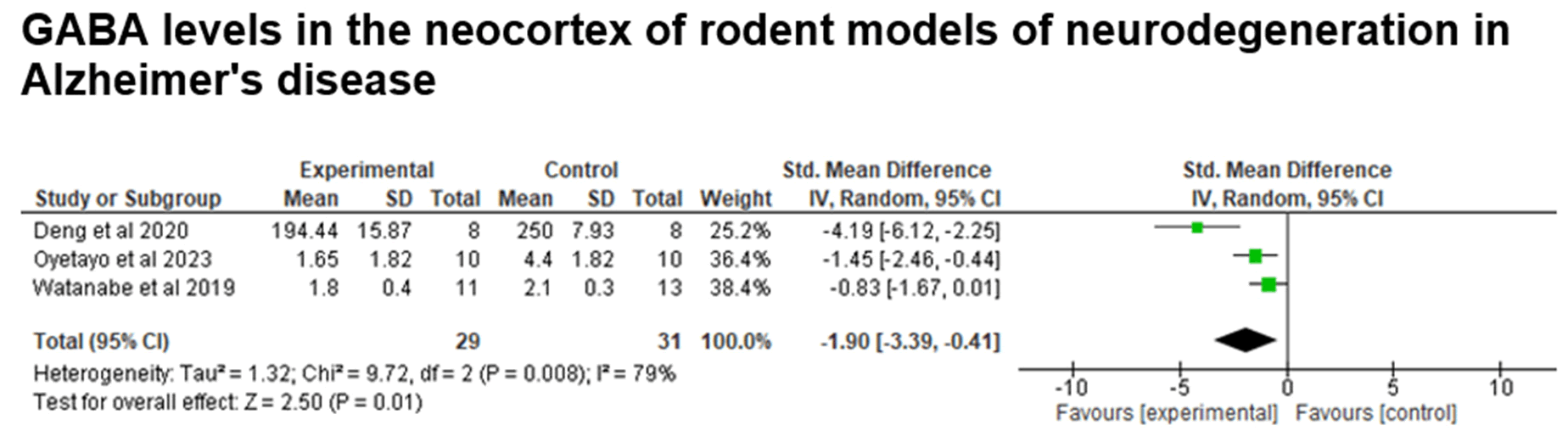
Fig. 6. Data from the meta-analysis of studies grouped using the keywords “Brain and GABA and neurodegeneration and Alzheimer”. GABA levels in the neocortex samples of rodents. Test for overall effect, Z=2.50 (P=0.01).
The meta-analysis was performed with data from 3 articles (3 studies), comparing GABA levels in experimental models of Alzheimer's disease between the control and experimental groups, with the rodent animal model. There is a significant difference between the groups, since the p-value is less than 0.05 (Z=2.50; p-value=0.01), indicating that GABA levels are lower in the neocortex in affected individuals (experimental group) (Figure 7).

Fig. 7. Data from the meta-analysis of studies grouped using the keywords “Brain and GABA and neurodegeneration and Alzheimer”. GABA levels in the hippocampus samples of rodents. Test for overall effect, Z=2.50 (P=0.01).
The meta-analysis was performed using data from 7 articles (8 studies), comparing GABA levels in experimental models of Alzheimer's disease between control and experimental groups, with the animal model being rodents. There is a significant difference between the groups, since the p-value is less than 0.05 (Z=3.91; p-value<0.0001), indicating that GABA levels are lower in the hippocampus in affected individuals (experimental group) (Figure 8).

Fig. 8. Data from the meta-analysis of studies grouped using the keywords “Brain and GABA and neurodegeneration and Alzheimer”. GABA-A receptor expression in the neocortex samples of rodents. Test for overall effect, Z=2.47 (P=0.01).
The meta-analysis was performed with data obtained from 2 articles (6 studies), comparing GABA-A receptor expression in experimental models of Alzheimer's disease between the control and experimental groups, considering the rodent and neocortex animal models. There is a significant difference between the groups, since the p-value is less than 0.05 (Z=2.47; p-value=0.01), indicating that GABA-A expression is higher in the neocortex in affected individuals (Figure 9).

Fig. 9. Data from the meta-analysis of studies grouped using the keywords “Brain and GABA and neurodegeneration and Alzheimer”. GABA-A receptor expression in the hippocampus samples of rodents. Test for overall effect, Z=2.49 (P=0.0.1).
The meta-analysis was performed with data obtained from 3 articles (8 studies), comparing GABA-A receptor expression in experimental models of Alzheimer's disease between the control and experimental groups, considering the animal model of rodents and the hippocampal region. There is a significant difference between the groups, since the p-value is less than 0.05 (Z=2.49; p-value=0.01), indicating that GABA-A expression is higher in the hippocampus in affected individuals (Figure 10).

Fig. 10. Data from the meta-analysis of studies grouped using the keywords “Brain and GABA and neurodegeneration and Alzheimer”. vGAT levels in the hippocampus samples of rodents. Test for overall effect, Z=0.94 (P=0.35).
The meta-analysis was performed with data from 2 articles (2 studies), comparing the vGAT transporter in experimental models of Alzheimer's disease between the control and experimental groups, being the rodent animal model. There is no evidence of a difference between the groups in the hippocampus, since the p-value is greater than 0.05 (Z=0.94; p-value=0.35) and the confidence interval includes zero (Figure 11).

Fig. 11. Data from the meta-analysis of studies grouped using the keywords “Brain and GABA and neurodegeneration and Parkinson”. GABA levels in the striatum samples of rodents. Test for overall effect, Z=2.53 (P=0.01).
The meta-analysis was performed with data from 14 articles (15 studies), comparing GABA levels in experimental models of Parkinson's disease between the control and experimental groups, with the animal model being rodents. There is a significant difference between the groups, since the p-value is less than 0.05 (Z=2.53; p-value =0.01), indicating that GABA levels are lower in the striatum in affected individuals (experimental group) (Figure 12).
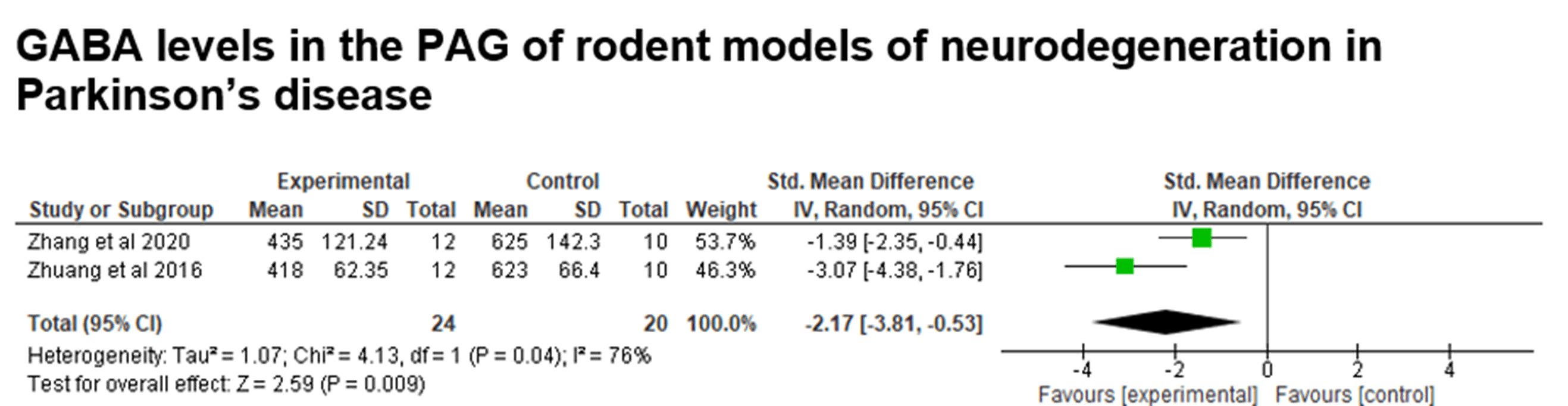
Fig. 12. Data from the meta-analysis of studies grouped using the keywords “Brain and GABA and neurodegeneration and Parkinson”. GABA levels in the PAG samples of rodents. Test for overall effect, Z=2.59 (P=0.009).
The meta-analysis was performed with data from 2 articles (2 studies), comparing GABA levels in experimental models of Parkinson's disease between the control and experimental groups, with the rodent animal model. There is a significant difference between the groups, since the p-value is less than 0.05 (Z=2.59; p-value=0.009), indicating that GABA levels are lower in the periaqueductal gray matter in affected individuals (experimental group) (Figure 13).

Fig. 13. Data from the meta-analysis of studies grouped using the keywords “GABA and Alzheimer and cortex”. GABA levels in the neocortex post-mortem samples. Test for overall effect, Z=3.49 (P=0.0005).
The meta-analysis was performed with data obtained from 2 articles (5 studies), comparing GABA levels in Alzheimer's disease between the control and experimental groups (human patients). There is a significant difference between the groups, since the p-value is less than 0.05 (Z=3.49; p-value=0.0005), indicating that GABA levels are lower in the neocortex in affected individuals (Figure 14).

Fig. 14. Data from the meta-analysis of studies grouped using the keywords “GABA and Alzheimer and Cortex”. GABA levels in the neocortex post-mortem samples. Test for overall effect, Z=1.02 (P=0.031).
The meta-analysis was performed with data from 4 articles (6 studies), comparing GABA levels in Parkinson's disease between the control and experimental groups (human patients). There is no evidence of a difference between the groups in the neocortex, since the p-value is greater than 0.05 (Z=1.02; p-value=0.31) and the confidence interval includes zero (Figure 15).

Fig. 15. Data from the meta-analysis of studies grouped using the keywords “Glutamate and cortex and Alzheimer”. Glutamate levels in the neocortex samples of rodents. Test for overall effect, Z=1.27 (P=0.20).
The meta-analysis was performed with data from 4 articles (4 studies), comparing glutamate levels in experimental models of Alzheimer's disease between the control and experimental groups, with the rodent animal model. There is no evidence of a difference between the groups in the cortex, since the p-value is greater than 0.05 (Z=1.27; p-value=0.20) and the confidence interval includes zero (Figure 16).

Fig. 16. Data from the meta-analysis of studies grouped using the keywords “Glutamate and hippocampus and Alzheimer”. Glutamate levels in the hippocampus samples of rodents. Test for overall effect, Z=1.24 (P=0.20).
The meta-analysis was performed with data from 7 articles (12 studies), comparing glutamate levels in experimental models of Alzheimer's disease between the control and experimental groups, with the rodent animal model. There is no evidence of a difference between the groups in the hippocampus, since the p-value is greater than 0.05 (Z=1.24; p-value=0.22) and the confidence interval includes zero (Figure 17).

Fig. 17. Data from the meta-analysis of studies grouped using the keywords “Glutamate and striatum and Parkinson”. Glutamate levels in the striatum samples of rodents. Test for overall effect, Z=1.69 (P=0.09).
The meta-analysis was performed with data from 10 articles (12 studies), comparing glutamate levels in experimental models of Parkinson's disease between the control and experimental groups, with the rodent animal model. There is no evidence of a difference between the groups in the striatum, since the p-value is greater than 0.05 (Z=1.69; p-value=0.09) and the confidence interval includes zero (Figure 18).
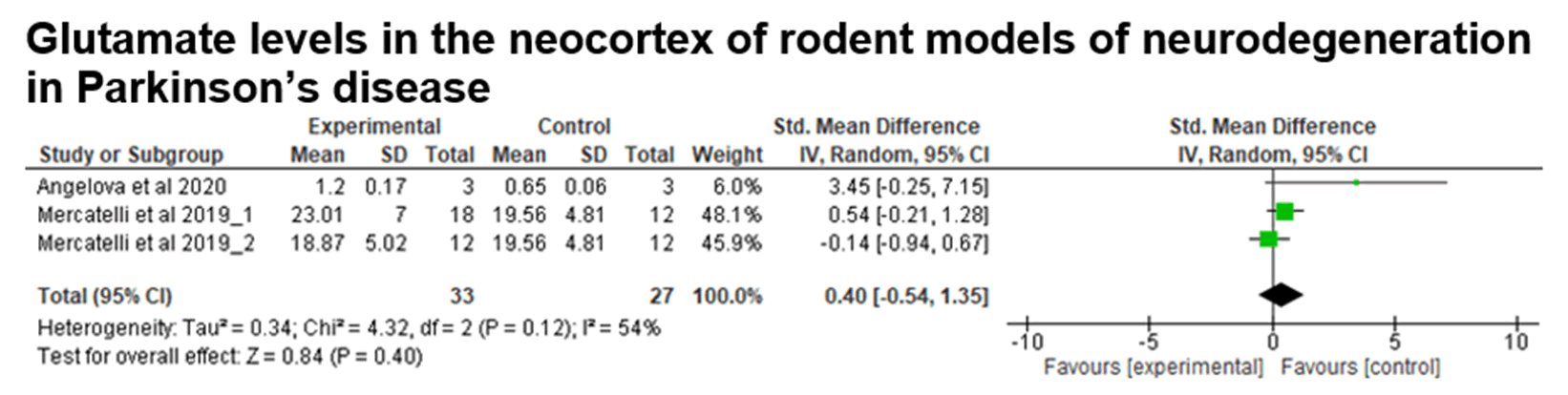
Fig. 18. Data from the meta-analysis of studies grouped using the keywords “Glutamate and neocortex and Parkinson”. Glutamate levels in the neocortex samples of rodents. Test for overall effect, Z=0.84 (P=0.40).
The meta-analysis was performed with data from 2 articles (3 studies), comparing glutamate levels in Parkinson's disease models between the control and experimental groups, with the rodent animal model. There is no evidence of a difference between the groups in the cortex, since the p-value is greater than 0.05 (Z=0.84; p-value=0.40) and the confidence interval includes zero.
Discussion
In order to answer contradictory questions about the involvement and importance of components of the neurochemical regulatory brain systems, GABAergic and glutamatergic, in the neurodegeneration process associated with Alzheimer's and Parkinson's diseases, we gathered and analyzed the data obtained from various studies published in the PubMed and Scopus databases between 1987 and 2024, in which the authors measured the molecular parameters of these two systems in samples obtained post-mortem from patients and in experimental models (rodents) of the two diseases. There is no systematic survey in the literature aimed at verifying the relevance and degree of significance of the relationships between these parameters and the phenomenon of neurodegeneration observed in these diseases.
As mentioned in the methods section, the study was divided into two stages. The first stage focused on neurodegenerative processes in a more general way. In this phase, studies were selected in which the measurements were carried out on total brain samples and therefore without specifying the region. The partial analysis of the data obtained in the first stage showed a predominance of studies dealing with Alzheimer's and Parkinson's disease and, also indicated significant effects related to GABAergic molecular components. To further explore this result, we decided to carry out the search for studies of central GABAergic parameters in these two diseases, adding to the keywords of the first stage, the specification of brain regions generally affected in these two conditions and found in studies selected in the first stage. This included the regions of the neocortex, hippocampus, striatum and PAG. The substantia nigra par compacta, which is important in PD, was not included in the 2nd stage because it did not appear in the studies selected in the 1st stage of the study. An extension of the study to include the substantia nigra would be very important as part of the planning of future studies.
The main findings of this study were that GABA levels in AD are significantly lower in the neocortex of rodents and humans and in the hippocampus of rodents. This decreased GABA was also observed in human whole brain samples. GABA-A receptor expression was higher in the neocortex and hippocampus of affected individuals (rodents). On the other hand, glutamate levels were not affected in either of these two regions, neocortex and hippocampus, in rodents.
In Parkinson's disease, GABA levels were lower in the striatum and periaqueductal grey matter (PAG) of affected individuals. Unlike AD, there was no difference in GABA levels in the neocortex in humans and we found no study of GABA levels in the hippocampus and neocortex of rodents for PD, using the criteria used. There were no differences in glutamate levels in the striatum and neocortex. No studies were found measuring glutamate levels in the PAG.
Analyses of the data from the studies selected in the first and second stages of the study showed that there were no differences between the control and experimental groups in glutamate levels in neurodegenerative processes, including Alzheimer's disease and Parkinson's disease, both in measurements taken in total brain samples and in specific brain areas, such as the neocortex and hippocampus in AD, and in the striatum and neocortex in PD. These findings suggest that glutamate somehow remains unchanged after the neurodegenerative process that takes place in these affected brain regions in AD (neocortex and hippocampus) and PD (neocortex and striatum). This is an interesting finding, as it suggests that the compensatory mechanisms for maintaining glutamate levels seem to be effective in re-establishing alterations induced by neuronal loss in diseases of different etiologies, such as AD and PD, as also happens with noxious stimuli (e.g. toxins, radiation, chemical products, stress), which could affect essential brain functions dependent on glutamatergic activity This finding of no change in glutamate levels, together with the data on decreased GABA levels, are the basis for the following considerations. It is known that an excessive increase in glutamate levels can lead to excitability of the glutamatergic system, resulting in the death of neurons and that the different buffering mechanisms (e.g., glia) and control of glutamate levels are important resources to avoid triggering this process that results in neuronal death. However, the data suggest that a decrease in GABA concentration, without a change in glutamate levels, may also represent an important component in the neurodegeneration process in these diseases. We therefore considered the following question: Would a decrease in GABA levels play an important role in neuronal death or would it be the consequence of a previous metabolic alteration, such as a reduction in Krebs cycle activity? A decrease in GABA levels could induce glutamatergic excitability independent of buffering mechanisms, since in this case there would be no absolute increase in glutamate levels. Another point that could be considered is that a decrease in GABA levels affects neurotrophic factors which could trigger a neurodegenerative process. We must also consider that the primary alteration may be in BDNF levels. During the neurodegenerative and neurogenerative process, there is a close interaction between BDNF and GABAergic transmission [18-20]. In inhibitory synapses, both increased and decreased BDNF levels contribute to a dysfunction of GABAergic transmission, resulting in altered GABA release and transport. According to this can disrupt the balance between excitatory/inhibitory neurotransmission, contributing to the underlying mechanisms of neurodegeneration.
However, the relationship between BDNF and GABAergic systems remains largely unknown; therefore, further studies investigating the precise interactions between BDNF and GABAergic transmission are needed. In this sense, the results presented here corroborate the importance of understanding these interactions. The GABAergic and BDNF systems are involved in essential brain functions, from neuronal development and the survival of neurons to learning and memory. In conclusion, the present meta-analysis corroborates the relevant role that the GABAergic inhibitory system may play in the mechanism of neurodegeneration in both AD and PD. Another possibility to be considered is that the decrease in GABA may be a consequence of neurodegeneration caused by the decrease in glucose oxidation by the Krebs cycle and, therefore, a reduction in the levels of the GABA precursor α-ketoglutarate and, also an impairment of glutamate levels. In this direction, it could be surmised that since glutamate is also a precursor of GABA and since this is impaired in the early stages of the process, it could be one of the important causes of the decrease in GABA. On the other hand, it is known that various neurodegenerative insults can induce excitotoxicity by increasing Glu. In this case, Glu could serve as a substrate for GABA replacement, through a mechanism that simultaneously re-establishes Glu levels, which would explain the absence of alterations in later stages of the process. However, this compensation doesn't seem to adequately meet the demand for GABA replacement, and if GABA is reduced, it could induce glutamatergic excitability, regardless of glutamate levels, which in this situation would be relatively high. Another explanation for the dysfunction of GABAergic neurons in individuals with AD may be a dysfunction in GABA synthesis or an enzyme deficiency in the glutamate-glutamine cycle, which may be impaired, with consequent toxicity leading to neuronal death. This study opens up perspectives for these hypotheses to be better tested.
Electrophysiological studies, measuring the activity of the glutamatergic and GABAergic systems in specific regions, associated with specific molecular markers at different stages of the neurodegenerative process, may help to clarify these issues. Theoretically, any oxidative dysfunction could affect α-ketoglutarate levels and imply a decrease in GABA levels, with a consequent increase in the system's excitability, regardless of glutamate levels. There is evidence of a relationship between glucose metabolic dysfunction and neurodegenerative diseases. In other words, any relative increases in glutamate levels that are detected at a stage before neuronal death could be a response to a primary dysfunction in glucose metabolism, specifically in the Krebs cycle or in α-ketoglutarate levels. Therefore, whether the decrease in GABA levels in the regions affected by neurodegeneration in PD (neocortex and striatum) and AD (neocortex and hippocampus) means cause’ or ‘consequence’ of a previous event needs to be better studied. An important observation from this review and meta-analysis is the agreement that similar phenomena occur in places that are affected differently in either of the two diseases. It is known that there are regional differences in the activity of glutamate decarboxylase in the brain, which is responsible for producing GABA from Glu.
Returning to the results relating to GABA levels, as mentioned above, the data obtained in AD show that there was a significant decrease in central GABA levels in the neocortex, both in studies in which the authors used rodents as a model (p=0.01) and in postmortem studies in humans (p=0.0005). The fact that data obtained from samples from clinical and experimental studies in rodents, and therefore with different sample characteristics are in agreement reinforces the importance of this finding.
Although the neocortex is an impaired region in PD, there was no difference in GABA levels in this region in studies using post-mortem samples in humans, suggesting a possible functional difference between AD and PD in the neocortex in the findings relating to this parameter. One possible explanation for this difference could be the neuropathological impairment observed in the neocortex of AD patients, which is quantitatively greater compared to PD. For example, the presence of a significant amount of senile plaques and neurofibrillary tangles in the neocortex region in AD. Although there is evidence of the presence of neurofibrillary tangles in PD, the quantitative characteristics differ from AD. It is known that beta-amyloid peptide toxicity contributes to GABAergic neuron dysfunction, followed in later stages by neuronal death.
In addition, showed that GABA levels were significantly reduced in the experimental AD group and the authors' conclusion is that the accumulation of tau protein (neurofibrillary tangles) in the CA3 region of the hippocampus could damage GABAergic neurons directly or compromise GABAergic functions indirectly by affecting the synaptic connections between pyramidal neurons and GABAergic neurons. A similar phenomenon could also occur in the neocortex. The accumulation of tau protein selectively reduced GABA levels among various monoamines and neurotransmitters evaluated.
Another explanation for not detecting changes in GABA levels in the neocortex in PD is the broad spectrum of PD, with conditions in which different brain regions and functions are or are not compromised. In this case, it would be important to assess neocortical GABA levels by separating the subtypes of PD, such as cases in which cognitive dysfunction occurs. We can see that the group of patients diagnosed with Parkinson's and with hallucination symptoms show a decrease in GABA levels in the neocortex, while in patients without hallucination GABA levels are normal, indicating a diversity in the neurobiological bases of different subtypes of the disease. Areas of the temporal cortex and prefrontal cortex are related to episodes of hallucination.
As can be seen from the data presented here, in the literature there is an inconclusive overview of changes in GABA levels and GABAergic components in the brain areas of patients with AD and PD and, therefore, grouping the data using the meta-analysis method represents an important contribution of the present study.
In the literature, we found controversy regarding changes in the expression of the GABA-A receptor in the experimental model of AD. Evidence obtained in a study. Shows that there was no difference in GABA-A expression of the alpha1 and alpha5 subunits and an increase in the delta subunit. This indicates that the effect observed depends on the experimental model used. On the other hand, some authors have found alterations and suggest that the expression levels of the alpha 1 and alpha 5 subunits in the hippocampus are predictors of the severity of AD neuropathology. In the present study, the pooling of data obtained from different articles shows that GABA-A expression in AD is higher in affected individuals, in rodent studies, in the neocortex and hippocampus.
The increase in the GABA-A receptor is consistent with the finding of decreased levels of GABA in the neocortex in experimental models of AD and in humans and in the hippocampus in experimental models. It is known that a decrease in neurotransmitter levels in synapses induces a compensatory effect that culminates in an increase in receptor expression. Other evidence to explain this finding is that subunits of the GABA-A receptor are altered with age and in AD. In order to preserve hippocampal function, surviving hippocampal neurons increase the synthesis and expression of GABA-A receptor subunits in an attempt to preserve the activity of the inhibitory hippocampal circuitry, even under conditions of decreased GABA. This mechanism of compensatory increase is reported in the late stages of AD, where the neurodegeneration process is accentuated. This increase demonstrates the intrinsic plasticity of the adult brain even during the progression of the disease and the importance of the GABAergic inhibitory circuits in this region, which play a functional role in the individual's cognition.
Data on the expression of the GABA-A receptor for Parkinson's disease was not shown because no studies were found that met the inclusion and exclusion criteria established here, in the searches carried out with the keywords related to this topic.
No significant difference was observed between the groups in GABA levels and neurodegenerative processes, with data from the first stage, when considering studies in rodents, in whole brain measurements. On the other hand, a significant decrease in GABA levels was found in neurodegenerative processes in humans. The failure to detect this effect in rodents may be due to the wide variety of experimental models between the studies.
The data shows that despite the establishment of inclusion and exclusion criteria in the selection of articles, there is a high degree of heterogeneity in the meta-analysis groups. A possible explanation for this could be the collection of data at different stages of the disease or of the neurodegeneration process, in the case of the use of experimental models. Parkinson's disease is characterized by dopamine deficiency in the striatum. The data obtained here show that GABA levels are also decreased in this region and also in the PAG.
For AD, Shows data on the vesicular Gaba Transporter (vGAT) in the hippocampus. Despite the significant changes in GABA levels in the hippocampus, no change was observed in the levels of this transporter.
Conclusion
The finding indicating a difference in the impairment of GABA levels in the neocortex between AD and PD, and the failure to detect the same phenomenon in the hippocampus, gave rise to some questions, as set out below. Is this a neuropathological difference in the disease or just a lack of studies in the hippocampus of PD patients? A first step towards answering this question would be to carry out a review of the literature, broadening the keywords, focusing on components of the GABAergic system in the hippocampus of individuals with PD, separating the subtypes of disease and, in the case of rodents, characterizing the experimental models well. A relevant contribution of the data presented here is to encourage the design of new studies to answer the questions raised here. Thus, an important perspective is that future investigations of the role of GABAergic systems in regions not affected by neuronal loss in PD and AD may contribute to the understanding of important regulatory dysfunctions in less studied regions in these two diseases. The data presented here corroborate recent evidence that the original descriptions of neurodegenerative diseases as being mainly due to lesions in dopaminergic systems in the midbrain for PD and hippocampal cholinergic and glutamatergic lesions for AD and other dementias should be updated within a concept of multisystem neurodegeneration, where a progressive functional decline of the GABAergic system needs to be further considered and better studied.
References
- Nicola R, Okun E. Adult hippocampal neurogenesis: one lactate to rule them all. Neuromolecular Med. 2021;23:445-448.
[Crossref] [Google Scholar] [PubMed]
- Assaf Y. New dimensions for brain mapping. Science. 2018;362:994-995.
[Crossref] [Google Scholar] [PubMed]
- Nunes PT, Gómez-Mendoza DP, Rezende CP, et al. Thalamic proteome changes and behavioral impairments in thiamine-deficient rats. Neuroscience. 2018;385:181-197.
[Crossref] [Google Scholar] [PubMed]
- Romano IC, Ribeiro AM. Acquisition of a spatial navigation task in the padding pool induces an increase of GABA level in the hippocampus of Swiss mice. GSC Adv Res Rev. 2021;7:16-24.
[Crossref] [Google Scholar]
- de Sena IC. Learning a spatial navigation task alters GABA levels in the hippocampus of SWISS mice. 2015.
[Google Scholar]
- Pires RG, Pereira SR, Oliveira-Silva IF, et al. Cholinergic parameters and the retrieval of learned and re-learned spatial information: a study using a model of Wernicke–Korsakoff Syndrome. Behav Brain Res. 2005;162:11-21.
[Crossref] [Google Scholar] [PubMed]
- Vigil FA, de Fátima Oliveira-Silva I, Ferreira LF, et al. Spatial memory deficits and thalamic serotonergic metabolite change in thiamine deficient rats. Behav Brain Res. 2010;210:140-142.
[Crossref] [Google Scholar] [PubMed]
- Rego DG. Study of the relationships between age, spatial cognitive parameters and activity of the serotonergic system in the hippocampus of rats. 2014.
[Google Scholar]
- de Freitas-Silva DM, de Souza Resende L, Pereira SR, et al. Maternal thiamine restriction during lactation induces cognitive impairments and changes in glutamate and GABA concentrations in brain of rat offspring. Behav Brain Res. 2010;211:33-40.
[Crossref] [Google Scholar] [PubMed]
- Sun B, Halabisky B, Zhou Y, et al. Imbalance between GABAergic and glutamatergic transmission impairs adult neurogenesis in an animal model of Alzheimer's disease. Cell Stem Cell. 2009;5:624-633.
[Crossref] [Google Scholar] [PubMed]
- Taupin P. Neurogenesis in the adult central nervous system. C R Biol. 2006;329:465-475.
[Crossref] [Google Scholar] [PubMed]
- McNally GP, Augustyn KA, Richardson R. GABAA receptors determine the temporal dynamics of memory retention. Learn Mem. 2008;15:106-111.
[Crossref] [Google Scholar] [PubMed]
- Toso L, Johnson A, Bissell S, et al. Understanding the mechanism of learning enhancement: NMDA and GABA receptor expression. Am J Obstet Gynecol. 2007;197:267-e1.
[Crossref] [Google Scholar] [PubMed]
- Bazzigaluppi P, Beckett TL, Koletar MM, et al. Earlyâ?stage attenuation of phaseâ?amplitude coupling in the hippocampus and medial prefrontal cortex in a transgenic rat model of Alzheimer's disease. J Neurochem. 2018;144:669-679.
[Crossref] [Google Scholar] [PubMed]
- Rissman RA, de Blas AL, Armstrong DM. GABAA receptors in aging and Alzheimer’s disease. J Neurochem. 2007;103:1285-1292.
[Crossref] [Google Scholar] [PubMed]
- Forman SA, Chou J, Strichartz GR, et al. Farmacologia da neurotransmissão GABAérgica e glutamatérgica. 2009:148-165.
[Google Scholar]
- Kim J, Lee S, Kang S, et al. Brain-derived neurotropic factor and GABAergic transmission in neurodegeneration and neuroregeneration. Neural Regen Res. 2017;12:1733-1741.
[Crossref] [Google Scholar] [PubMed]
- Levine ES, Dreyfus CF, Black IB, et al. Brain-derived neurotrophic factor rapidly enhances synaptic transmission in hippocampal neurons via postsynaptic tyrosine kinase receptors. Proc Natl Acad Sci USA. 1995;92:8074-8077.
[Crossref] [Google Scholar] [PubMed]
- Ganguly K, Schinder AF, Wong ST, et al. GABA itself promotes the developmental switch of neuronal GABAergic responses from excitation to inhibition. Cell. 2001;105:521-532.
[Crossref] [Google Scholar] [PubMed]
- Andrews-Zwilling Y, Bien-Ly N, Xu Q, et al. Apolipoprotein E4 causes age-and Tau-dependent impairment of GABAergic interneurons, leading to learning and memory deficits in mice. J Neurosci. 2010;30:13707-13717.
[Crossref] [Google Scholar] [PubMed]























