Case Report - (2022) Volume 13, Issue 4
ISAAC syndrome
Sumeet Swapan Roy*,
Raghav Kapoor,
Rahul Saini and
Nikita Goel
Department of Neurology, Max Hospital Saket, New Delhi, India
*Correspondence:
Sumeet Swapan Roy, Department of Neurology, Max Hospital Saket,
New Delhi,
India,
Email:
Received: 08-Apr-2022, Manuscript No. ipjnn-22-12720;
Editor assigned: 10-Apr-2022, Pre QC No. P-12720;
Reviewed: 20-Apr-2022, QC No. Q-12720;
Revised: 23-Apr-2022, Manuscript No. R-12720;
Published:
30-Apr-2022
Introduction
ISSAC Syndrome is a rare acquired autoimmune disorder
characterized by peripheral nerve hyperexcitability in form
of spontaneous continuous skeletal muscle over activity
presenting as twitching and painful cramps it is often
associated with stiffness, pseudomyotonia, pseudotetany
and weakness [1,2]. Patients are commonly treated with
symptomatic therapies such as carbamazepine, valproate,
phenytoin with no clinical improvement [3,4]. While
immunomodulatory approaches are still the mainstay for
the treatment, no clinical trial is available to date and specific
management is unknown. A pathology of antibody mediated
autoimmune mechanisms directed against peripheral nerve
voltage gated potassium channels (anti-VGKC antibody),
which regulate nerve excitability, is implicated [5]. There
are 60 percent of patients with Isaac syndrome having no
defined antibody target [4].
Here we are reporting a case of a CASPR 2 and LGI 1
antibody positive patient who presented with continuous
muscle twitch on his lower limb and diffuse muscle pain.
He was treated and showed good clinical response to
immunomodulatory therapy.
Case Presentation
Our patient is a 34-year-old man who is admitted with
complaints of severe backache and paresthesia. However,
he had no decline in cognitive function or impairment of
higher mental functions. Examination showed fasciculations
on her bilateral lower limbs. Power was 5/5 and symmetrical
in all four limbs. Radiological investigations such as MRI
Brain and MRI Sacroiliac Joint showed no significant
abnormality and PET SCAN did not show any uptake. Nerve
Conduction Velocity revealed a normal study. Serological
studies including ANA vasculitis Pannel was negative.
Serum negative HLA B27 was Negative, while Autoimmune
Encephalitis panel showed LGI-1 antibody (VGKC type) as
well as CASPR2 antibody positivity. Electromyography was
done which showed continuous and spontaneous irregular
duplets and triplets from a single motor unit in bilateral
FDI, Vastus Lateralis, and right Gastrocnemius Muscle.
Electrophysiological study showed neurogenic pattern with
evidence of active denervation and reinnervation, suggestive
of anterior horn cell disease (Fig. 1).
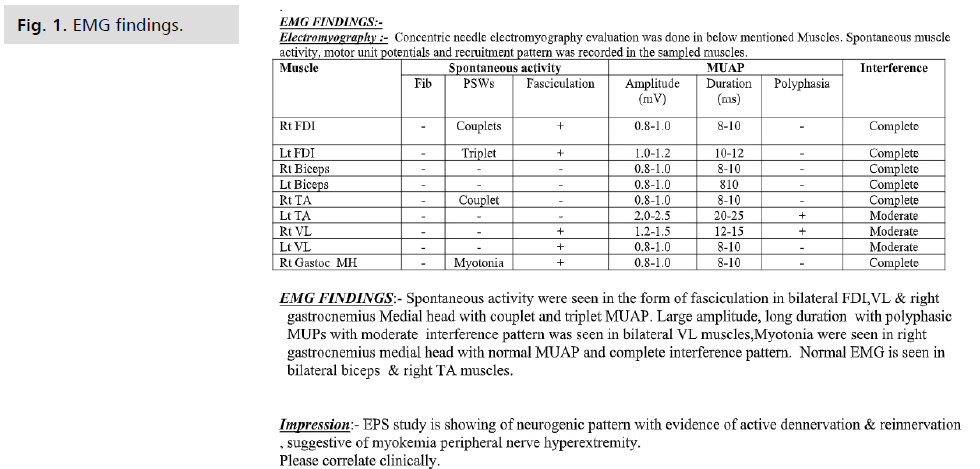
Fig 1: EMG findings.
After a few days of conservative management,
methylprednisolone therapy was initiated with a dose of 1 gram for three days, showing clinical improvement and
then again for 2 more days to a total of 5 days. It was then
switched to oral steroids that were tapered off in a span
of 2 weeks. Patient was also started on plasma exchange
therapy for 5 days on alternate days. After the full course
of treatment, the patient showed clinical improvement and
recovered significantly.
Discussion
Acquired neuromyotonia also known as ISSAC Syndrome
is described as persistent generalized muscle twitching and
stiffness associated with spontaneous motor unit action
potential on needle electromyograph. ISSAC Syndrome is a
peripheral nerve hyperexcitability syndrome of spontaneous
muscle fiber activity resulting in hyperexcitability of
peripheral nerve origin. It is reported to be twice as common
in males than females and the average age of onset is the
mid-40s.
The pathological Hallmark of ISSAC syndrome is the anti
VGKC complex antibody binding to LGI 1 and CASPR 2
which are the components of VGKC complex [6]. Among
these, LGI 1 is a neuronal protein which is expressed
predominantly in hippocampus and associates specifically
with Kv1.1 subunits in CNS presynaptic terminals [7]. LGI
1 is strongly associated with encephalitis, specifically limbic
encephalitis.
The CASPR 2 is a transmembrane protein which is required
for localization of Kv1.1 and 1.2 at the juxtaparanodes [8].
CASPR 2 antibodies are found in ISSAC syndrome or
Morvan’s Syndrome. Morvans Syndrome is a rare condition
which presents with neuromyotonia plus autonomic
disturbance and CNS involvement that includes insomnia
and confusion [9]. Our Patient is both CASPR 2 and LGI 1
positive and has no autonomic disturbance.
ISSAC Syndrome presents with muscle twitching, cramps,
hyperhidrosis, slow relaxation of muscle after muscle contraction [10]. Diagnosis can also be made by EMG
showing high intraburst of irregular frequency. The abnormal
EMG is characterized by doublet, triplet ad multiplet
single unit discharges which is also seen in our present case
[10,11]. These changes are also known as myokymic and
neuromyotonic discharges [11].
Clinically ISSAC syndrome patients show improvement in
symptoms with Immunosuppressive and Immunomodulatory
therapy, this improvement is reflected in the antibody levels
as well. In some reported cases the antibody levels become
undetected with the Immunomodulatory therapy [4].
ISSAC Syndrome is found to be associated with other
autoimmune diseases including myasthenia gravis and
crohn's disease. It is also associated with certain neoplasms,
of which thymoma is the major risk factor. A PET scan is
therefore may be sometimes required in case of doubt and no
signs and symptoms. Sometimes this syndrome is presented
years before the neoplasm is discovered [12-14]. Certain
studies have shown association of neoplasm to be as high as
31.6% with CASPR 2 antibody positivity [7].
Conclusion
Management of Issac Syndrome includes symptomatic
treatment at first with Carbamazepine, phenytoin,
sodium valproate, which can be used in combination
if necessary. Suggested treatment includes steroid pulse
dose along with plasmapheresis in cases not responding to
steroids alone. Refractory cases can be managed with an
immunomodulator’s, mainly azathioprine or methotrexate.
In some refractory cases, Rituximab showed to be effective.
In cases associated with neoplasms, improvement after
removal of an underlying neoplasm is reported.
Conflict of Interest
Authors declare there is no conflict of interest.
Funding
None.
Consent of Publication
Written consent is obtained for the paper.
REFERENCES
- Skeie GO, Apostolski S, Evoli A, et al. Guidelines for treatment of autoimmune neuromuscular transmission disorders. Eur J Neurol. 2010;17(7):893-902.
Google Scholar, Crossref, Indexed at
- Fleisher J, Richie M, Price R, et al. Acquired neuromyotonia heralding recurrent thymoma in myasthenia gravis. JAMA Neurol. 2013;70:1311-1314.
Google Scholar, Crossref, Indexed at
- Orsucci D, Cafforio G, Moscato G, et al. Steroids and plasma exchange in Isaacs' syndrome with anti-Caspr2 antibodies. Neuroimmunol Neuroinflammation. 2018;5(3):7.
Google Scholar, Crossref, Indexed at
- Song J, Jing S, Quan C, et al. Isaacs syndrome with CASPR2 antibody: A series of three cases. J Clin Neurosci. 2017;41:63-66.
Google Scholar, Crossref, Indexed at
- Irani SR, Alexander S, Waters P, et al. Antibodies to Kv1 potassium channelcomplex proteins leucine-rich, glioma inactivated 1 protein and contactinassociated protein-2 in limbic encephalitis, Morvan’s syndrome and acquired neuromyotonia. Brain. 2010;133(9):2734-2748.
Google Scholar, Crossref, Indexed at
- Arimura K, Sonoda Y, Watanabe O, et al. Isaacs’ syndrome as a potassium channelopathy of the nerve. Muscle Nerve Suppl. 2002;11:S55-S58.
Google Scholar, Crossref
- Lai M, Huijbers MG, Lancaster E, et al. Investigation of LGI1 as the antigen in limbic encephalitis previously attributed to potassium channels: a case series. Lancet Neurol. 2010;9(8):776-785.
Google Scholar, Crossref, Indexed at
- Irani SR, Vincent A. Voltage-gated potassium channel-complex autoimmunity and associated clinical syndromes. Handb Clin Neurol. 2016;133:185-197.
Google Scholar, Crossref, Indexed at
- Klein CJ, Lennon VA, Aston PA, et al. Insights from LGI1 and CASPR2 potassium channel complex autoantibody subtyping. JAMA Neurol. 2013;70:229-234.
Google Scholar, Crossref, Indexed at
- Ahmed A, Simmons Z. Isaacs syndrome: a review. Muscle Nerve. 2015;52(1):5-12.
Google Scholar, Crossref
- Rana SS, Ramanathan RS, Small G, et al. Paraneoplastic Isaacs’ syndrome: a case series and review of the literature. J Clin Neuromuscul Dis. 2012;13(4):228-233.
Google Scholar, Crossref, Indexed at
- Magnuson RL, Abrams BM, Reiss JE. Continuous muscle fiber activity: IsaacsMertens’ syndrome. Arch Phys Med Rehabil. 1972;53:282-285.
- Hart IK, Maddison P, Newsom-Davis J, et al. Phenotypic variants of autoimmune peripheral nerve hyperexcitability. Brain. 2002;125:1887-1895.
Google Scholar, Crossref, Indexed at
- Hart IK, Waters C, Vincent A, et al. Autoantibodies detected to expressed K+ channels are implicated in neuromyotonia. Ann Neurol. 1997;41(2):238-246.
Google Scholar, Crossref, Indexed at






